
This article defines pharmaceutical logistics, explains the core components of the pharma supply chain, examines the most pressing challenges organizations face today, and presents actionable solutions to address them.
TLDR:
- Pharmaceutical logistics requires strict temperature control, regulatory compliance, and chain-of-custody documentation that standard freight doesn't
- Cold chain failures waste up to 50% of global vaccines; maintaining 2–8°C integrity throughout transit is mission-critical
- DSCSA now mandates package-level electronic tracing using EPCIS standards; stabilization period ended November 2024
- At least 1 in 10 medicines in low- and middle-income countries are substandard or falsified, costing $30.5 billion annually
- Working with FDA-registered, GDP-compliant distributors dramatically reduces regulatory risk and ensures product authenticity
What Is Pharmaceutical Logistics?
Pharmaceutical logistics is the specialized management of storage, handling, and transportation of pharmaceutical products—including drugs, biologics, vaccines, and excipients—under conditions that preserve their safety, efficacy, and regulatory compliance. The European Medicines Agency (EMA) defines Good Distribution Practice (GDP) as the minimum standards that wholesale distributors must meet to ensure quality and integrity throughout the supply chain.
Several factors set pharma logistics apart from standard freight and distribution:
- Temperature requirements: Many products require 2–8°C refrigeration or below -20°C frozen storage; cold-chain medicines accounted for 38% of pharmaceutical market share in 2024 despite representing just 0.2% of global volume
- Documentation obligations: Every shipment requires batch records, certificates of analysis, serialization data, and chain-of-custody documentation
- Regulatory oversight: FDA 21 CFR Part 211, EU GDP guidelines, and country-specific import/export licensing create overlapping compliance requirements
- Stakes: Failures don't just cause financial loss—they render products ineffective or dangerous to patients
That combination of regulatory complexity and product sensitivity affects every organization that moves pharmaceuticals through the supply chain:
- Hospitals and retail pharmacies rely on uninterrupted product availability for patient care
- Clinical trial sponsors and research organizations face halted studies if shipments are delayed
- Vaccine developers and pharmaceutical manufacturers risk product loss from a single temperature excursion
Key Components of a Pharmaceutical Supply Chain
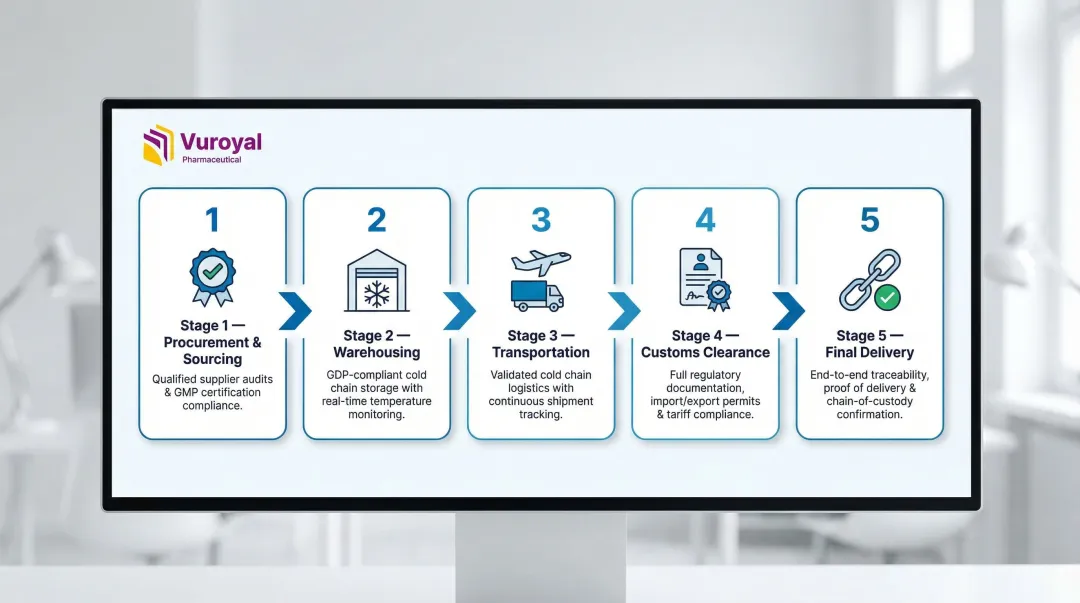
A compliant pharmaceutical supply chain integrates multiple specialized functions:
Core operational functions:
- Procurement and sourcing of raw materials or finished drugs from verified, audited suppliers
- Warehousing under controlled conditions with documented temperature mapping and continuous monitoring
- Transportation (domestic and cross-border) using qualified carriers and validated packaging
- Customs clearance for imports/exports, including drug business licenses and registration filings
- Final delivery to end users with chain-of-custody documentation and temperature verification
Good Distribution Practice (GDP): The standard governing pharmaceutical storage and distribution. Two key regulatory frameworks define compliance requirements:
- EU GDP guidelines (2013/C 343/01) set minimum standards for wholesale distributors across member states
- WHO Model Guidance (Annex 9) specifies operational requirements: temperature mapping, continuous monitoring (minimum six readings per hour), and facility qualification
Documentation and traceability requirements:
- Batch records and certificates of analysis proving product specifications
- Import/export permits and customs declarations
- Serialization data under the Drug Supply Chain Security Act (DSCSA)
- Chain-of-custody documentation tracking every transfer
Regulators expect continuous, calibrated monitoring and fully documented facility qualification. Failure to produce these records during inspection constitutes a critical GDP violation.
Top Challenges in Pharmaceutical Logistics
Maintaining Cold Chain Integrity
Many pharmaceuticals—including biologics, vaccines, and vaccine adjuvants—require strict temperature ranges throughout the entire journey from manufacturer to end user. Standard regulatory ranges include 2–8°C for refrigerated products, below -15°C or -20°C for frozen biologics, and -80°C ultra-low temperatures for certain mRNA vaccines.
Any temperature excursion can render products unsafe or ineffective. The WHO estimates that up to 50% of global vaccines are wasted due to cold chain failures, resulting in sub-potent vaccines that fail to provide adequate protection against targeted diseases.
Cold chain failures occur at multiple points:
- During loading/unloading when products move between controlled environments
- In transit across climate zones without adequate thermal protection
- At customs checkpoints where delays expose shipments to ambient temperatures
- Due to equipment failure in storage facilities or transport vehicles
The financial impact is severe. Cold-chain medicines made up $384 billion (32%) of total pharmaceutical value in 2021, despite accounting for just 0.2% of global volume. This extreme value density means logistics failures destroy high-value inventory and compromise patient outcomes.
Navigating Complex Regulatory Requirements
Pharmaceutical logistics operates under overlapping national and international regulations, each with distinct requirements:
- United States: Distributors must comply with FDA 21 CFR Part 211 for storage and handling and DSCSA for package-level traceability
- European Union: Wholesale distribution authorisation and full EU GDP guideline compliance are mandatory
- China: The NMPA now requires overseas Marketing Authorization Holders to designate a Domestic Responsible Person by July 1, 2025

Cross-border pharmaceutical shipments face additional complexity:
- Customs declaration requirements vary by country and product type
- Drug business licenses and registration filings differ across jurisdictions
- Import/export licensing regimes change frequently
- Documentation standards (certificates of origin, packing lists, bills of lading) must meet multiple authorities' requirements
Non-compliance can result in shipment seizures, product recalls, or loss of distribution licenses.
One critical deadline has already passed: following the November 27, 2023 DSCSA milestone, trading partners must exchange transaction data electronically and include package-level product identifiers. The 1-year stabilization period ended November 27, 2024, making full electronic compliance mandatory.
Counterfeit and Substandard Product Risk
The global pharmaceutical supply chain is vulnerable to infiltration of counterfeit, adulterated, or substandard drugs. The WHO estimates that at least 1 in 10 medicines in low- and middle-income countries are substandard or falsified, costing an estimated $30.5 billion annually.
The human cost is devastating: Renschler et al. (2015) estimated that 122,350 under-five malaria deaths in sub-Saharan Africa were associated with consumption of poor-quality antimalarials. This risk grows when organizations source from unverified distributors without proper licensing or quality certifications.
For specialty substances—vaccine excipients, reference listed drugs, biologics—authenticity directly affects research outcomes and patient safety. A counterfeit adjuvant introduced into a clinical trial can invalidate months of accumulated data. For generic drug developers, a substandard reference drug doesn't just delay timelines—it can collapse an entire development program.
Supply Chain Disruptions and Drug Shortages
Counterfeit risk targets product quality; supply chain disruptions threaten availability altogether. Pharmaceutical supply chains remain highly vulnerable to both. The FDA reported 55 new drug shortages in 2023, with 98 ongoing from previous years, while successfully preventing 236. Active shortages peaked at 323 in Q1 2024 before declining to 271.
Geographic concentration drives fragility: India holds 48% of all active API Drug Master Files for U.S. medicines, while China surpassed India in 2024 to hold 45% of new API DMF filings. Over-reliance on concentrated sources leaves supply chains vulnerable to geopolitical shocks, manufacturing delays, raw material shortages, and global health crises.
The downstream effects reach patients directly. An unavailable oncology drug forces protocol changes mid-treatment; a delayed vaccine adjuvant can put an entire clinical trial on hold, pushing back timelines by months.
Last-Mile Delivery for Specialty Products
The final mile of pharmaceutical delivery presents unique challenges, particularly for specialty drugs and clinical trial materials. The global clinical trial supply and logistics market was valued at $4,287.8 million in 2024 and is projected to reach $6,722.9 million by 2030.
Decentralized clinical trials (DCTs) and direct-to-patient (DTP) models add strict chain-of-custody and temperature constraints to last-mile delivery. Small-batch shipments must reach hospitals, clinics, or research labs on time and in condition—often requiring specialized couriers, real-time monitoring, and coordination with facility receiving protocols.
The cost of failure at this stage is measurable. A survey of nurses found that more than half had to reschedule at least one patient procedure in the past year due to a medical courier error, with each delayed procedure costing an average of $4,500 in idle operating room time.

Common last-mile failure points include:
- Courier errors causing missed or damaged deliveries
- Temperature excursions during final-leg transport without real-time monitoring
- Receiving protocol mismatches between shipper documentation and facility requirements
- Chain-of-custody gaps that trigger protocol deviations in clinical trials
For trial sponsors, even a single delivery failure can compromise data integrity and trigger costly protocol amendments.
Solutions to Overcome Pharmaceutical Logistics Challenges
Advanced Cold Chain Monitoring and Technology
IoT-enabled data loggers, real-time GPS temperature monitoring, and automated alert systems allow logistics teams to detect and respond to temperature excursions before products are compromised. The global cold chain monitoring market was estimated at $35.03 billion in 2024, reflecting the industry's investment in these technologies.
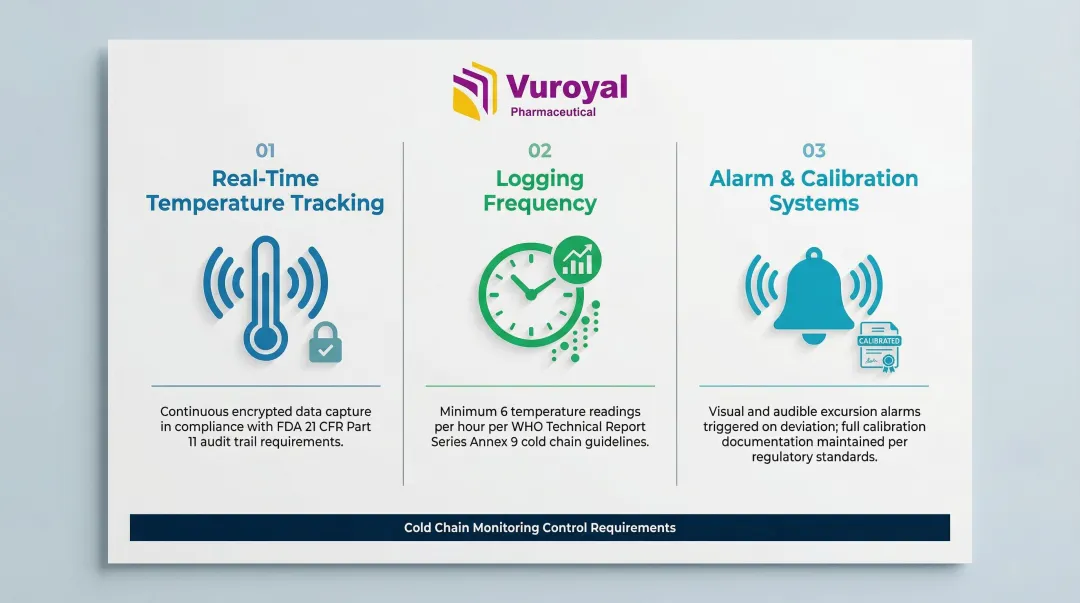
Key monitoring controls:
- Track shipment temperature in real time with encrypted electronic records compliant with FDA 21 CFR Part 11
- Log at least six readings per hour for storage and transport, as mandated by WHO Annex 9
- Trigger visual and audible alarms for temperature excursions, supported by calibration logs and testing documentation
Technology alone isn't sufficient—it must be paired with validated cold-storage facilities at each node of the supply chain. Facilities must complete temperature mapping to demonstrate air temperature profiles when empty and loaded, define zones not suitable for storage, and document the time taken for temperatures to exceed limits during power failure.
Partnering with Certified, Compliant Distributors
Working with an FDA-registered, licensed pharmaceutical wholesaler who holds drug business licenses, quality management certifications, and export/import qualifications significantly reduces regulatory and quality risk.
VuRoyal Pharmaceutical exemplifies this approach. As an FDA-registered, Massachusetts-approved wholesaler with drug business quality management certification and customs declaration registration, VuRoyal provides end-to-end supply chain capabilities for research organizations, clinical trial sponsors, and healthcare providers. The company maintains full compliance with:
- U.S. Pharmacopeia (USP) guidelines
- Title 21 CFR and the Prescription Drug Marketing Act (PDMA)
- Drug Supply Chain Security Act (DSCSA)
- Third-party compliance solutions for ongoing state and federal adherence
For organizations sourcing specialty products—such as vaccine adjuvants or reference listed drugs—partnering with distributors who hold multi-jurisdictional licenses and can navigate foreign regulatory systems eliminates major operational burdens.
Serialization and Track-and-Trace Systems
Product serialization assigns unique identifiers to each unit, enabling full supply chain visibility. DSCSA now requires trading partners to exchange transaction information that includes the product identifier at the package level, with the FDA strongly recommending the GS1 EPCIS standard for interoperable data exchange.
Benefits of serialization systems:
- Detect counterfeit products by verifying package-level identifiers against manufacturer records
- Comply with DSCSA requirements and avoid enforcement actions
- Execute rapid recalls by tracing affected lots to specific distribution points
- Protect confidential commercial information while enabling regulatory oversight
If a product is determined to be illegitimate, trading partners must notify the FDA within 24 hours using Form FDA 3911 and quarantine the product to prevent further distribution.
Diversified Sourcing and Single-Source Expertise
Diversifying supplier networks reduces dependency on single manufacturers or geographies. The trade-off is real, though: more suppliers means more coordination complexity and vendor management overhead.
A practical middle ground pairs diversified sourcing with a trusted single-source partner who combines procurement expertise, regulatory knowledge, and logistics capabilities. This model preserves supply resilience while reducing errors and administrative load.
VuRoyal's single-source model covers this end-to-end:
- Procurement and regulatory support, including imported drug approval registration and customs filing
- Customs clearance and specialized cold chain transportation
- Research-grade quality standards for clinical trial sponsors and research organizations
Regulatory Guidance and Customs Clearance Support
Specialized distributors who offer proactive regulatory guidance—including assistance with drug approval registration, customs declaration filing, and import licensing—free organizations from navigating complex compliance requirements without in-house regulatory expertise.
For international procurement, this support is essential. China's new NMPA requirements mandate that overseas Marketing Authorization Holders designate a Domestic Responsible Person by July 1, 2025, with joint liability for quality and safety. Navigating these evolving requirements requires distributors with established regulatory relationships and proven compliance track records.
The Role of Regulatory Compliance in Pharma Logistics
Regulatory compliance defines who can operate in pharmaceutical logistics — and under what conditions. The primary frameworks include:
United States: FDA 21 CFR Part 211 for storage and handling, DSCSA for package-level traceability, and state-level wholesale distribution licensing. Wholesale drug distributors and third-party logistics providers must report licensure information to the FDA annually.
European Union: Wholesale distribution authorisation issued by national competent authorities, with mandatory compliance with EU GDP guidelines. Competent authorities inspect distributors and log compliance statements in the publicly accessible EudraGMDP database.
Country-specific requirements: Import/export licensing, drug business permits, and registration filings that vary by jurisdiction and product type.
Non-compliance carries consequences well beyond financial penalties:
- Products can be quarantined at ports, halting distribution
- Distribution licenses can be revoked, shutting down operations
- Patient safety can be directly compromised, creating liability exposure
- Under FDA 21 CFR 211.208, products subjected to improper storage cannot be salvaged and returned to the marketplace unless laboratory evidence proves they meet all standards
These risks make regulatory standing a direct operational concern — not just a legal one. Organizations engaged in international pharmaceutical procurement should prioritize partners who already hold multi-jurisdictional licenses and can navigate foreign regulatory systems on their behalf.
Frequently Asked Questions
What is pharma logistics?
Pharma logistics refers to the specialized management of storing, handling, and transporting pharmaceutical products under conditions that preserve their safety and efficacy, governed by strict regulatory standards like GDP and FDA requirements.
What makes pharmaceutical logistics different from regular logistics?
Pharmaceutical logistics requires strict temperature control (often 2–8°C or below -20°C), regulatory compliance with GDP and FDA standards, detailed chain-of-custody documentation, and operates under life-critical stakes where failures can harm patients.
What is cold chain logistics in pharmaceuticals?
Cold chain logistics refers to the temperature-controlled supply chain required for products like vaccines, biologics, and certain drugs—maintaining defined temperature ranges from manufacturer to end user to preserve product efficacy and safety.
What regulations govern pharmaceutical logistics?
Key frameworks include FDA regulations and DSCSA in the U.S., WHO and EU GDP guidelines internationally, and country-specific drug import/export licensing requirements. For cross-border shipments, complying with the destination country's framework—not just the origin country's—typically governs clearance and approval.
How can organizations reduce pharmaceutical supply chain disruptions?
Several strategies reduce exposure to supply chain disruptions:
- Diversify sourcing across multiple geographies to avoid single-point failures
- Work with certified distributors who hold proper licenses and GDP compliance
- Invest in real-time visibility tools to catch delays before they escalate
- Maintain buffer inventory for critical or hard-to-source products
What licenses should a pharmaceutical distributor hold?
Core credentials to verify include:
- FDA registration as a pharmaceutical wholesaler
- State-level wholesale distribution licenses (requirements vary by state)
- Drug business quality management certification
- Customs declaration registration for cross-border operations


