
Introduction
In March 2020, supply chain failures turned into patient crises almost overnight. Three scenarios capture why healthcare logistics carries stakes no other industry can match:
- PPE shortages left hospital workers across the U.S. without basic protection during the pandemic's first wave
- A delayed shipment of a temperature-sensitive cancer drug forced oncology patients to postpone treatment
- A clinical trial for a promising vaccine stalled when the required adjuvant sat in customs limbo over missing regulatory documentation
When the medical supply chain breaks down, the consequences land directly on patients and researchers — not just balance sheets.
Healthcare logistics is the specialized discipline of managing how medical products move from manufacturers to the patients and researchers who need them.
This guide covers how the medical supply chain operates from sourcing to delivery, the key challenges facing the industry, and what to look for in a supply chain partner. Whether you're a hospital procurement director, research organization, pharmacy, or pharmaceutical manufacturer, these fundamentals determine whether the right product reaches the right place — on time and in compliance.
TLDR
- Healthcare logistics manages the end-to-end movement of pharmaceuticals, equipment, and supplies from manufacturer to patient
- The medical supply chain involves five stages: manufacturing, procurement, transportation, warehousing, and distribution
- Cold chain failures and compliance gaps drive most pharmaceutical supply disruptions — especially for biologics and specialty drugs
- Key challenges include supply disruptions, counterfeit drug risks, and complex international regulations
- For specialty and imported medicines, work with FDA-registered distributors offering end-to-end supply chain capabilities
What Is Healthcare Logistics?
Healthcare logistics is the specialized discipline of planning, coordinating, and executing the flow of medical products — pharmaceuticals, devices, and clinical supplies — from origin to end user. Unlike general commercial freight, healthcare supply chains demand far stricter precision at every step. Errors or delays don't just inconvenience customers; they can render products unsafe or ineffective, directly compromising patient outcomes.
The stakes are measurable: drug shortages cost U.S. hospitals nearly $900 million and 20 million labor hours annually, with 43% of facilities reporting medication errors directly related to supply chain failures. When a pharmaceutical distributor fails to maintain proper temperature controls during transport, an entire shipment of vaccines may become unusable. When customs documentation is incomplete, life-saving specialty drugs sit in regulatory limbo while patients wait.
Subcategories of Healthcare Products
Healthcare logistics encompasses three main product categories, each with distinct handling, storage, and regulatory requirements:
Pharmaceutical substances include chemical and biological agents used for treatment or research — from small-molecule drugs to complex biologics like monoclonal antibodies and vaccine adjuvants. Strict temperature control and narrow expiration windows make these the most logistically demanding products in the supply chain.
Medical equipment covers devices and machinery used in diagnostics and procedures, including imaging systems, surgical instruments, and monitoring devices. Specialized handling and sterility maintenance are non-negotiable requirements.
Medical supplies — gloves, syringes, dressings, diagnostic test kits — are high-turnover consumables. Their individual handling requirements are simpler than pharmaceuticals, but the sheer volume and clinical dependency make consistent availability critical.
How Healthcare Logistics Differs from General Logistics
Several key distinctions separate healthcare logistics from standard commercial freight:
- Temperature sensitivity requirements: Many pharmaceuticals must remain within 2-8°C throughout the supply chain, with even brief excursions rendering them ineffective
- Regulatory oversight: The FDA, DEA, state pharmacy boards, and international customs authorities impose strict licensing and documentation requirements
- Chain-of-custody documentation: Every transfer point requires verification, lot tracking, and temperature records
- Zero tolerance for delays: Unlike consumer goods, pharmaceutical delivery windows are measured in hours, not days
- Licensed distributor requirements: Only FDA-registered, state-licensed entities can legally handle prescription pharmaceuticals

Who Is Involved in the Healthcare Supply Chain?
The healthcare supply chain involves multiple interdependent stakeholders:
- Raw material suppliers provide active pharmaceutical ingredients (APIs) and excipients
- Pharmaceutical manufacturers produce finished drugs under GMP-certified conditions
- Licensed wholesalers and distributors bridge manufacturers and end users, handling procurement, verification, and logistics
- Hospitals and health systems receive and dispense medications to patients
- Pharmacies (retail and specialty) provide patient-level distribution
- Research organizations and clinical trial sponsors require reference drugs and specialty substances
- Regulatory authorities (FDA, DEA, state boards) enforce compliance and product safety
Each party's reliability directly affects whether the right product reaches the right patient at the right time. A single compliance failure at any point can disrupt the entire chain.
Why Healthcare Logistics Matters for Patient Outcomes
Healthcare logistics performance directly determines care quality. The right drug, at the right dose, at the right time — this formula depends entirely on supply chain execution.
Those labor hours — more than double the 2019 figure — represent a system under real strain. The clinical fallout is concrete:
- 27% of surveyed facilities reported direct patient care disruptions
- 41% saw outpatient infusion services affected
- 32% experienced delays to planned medical procedures
For time-sensitive treatments like chemotherapy or emergency medications, these aren't inconveniences. They're patient safety failures. When logistics break down, the consequences move far beyond the warehouse.
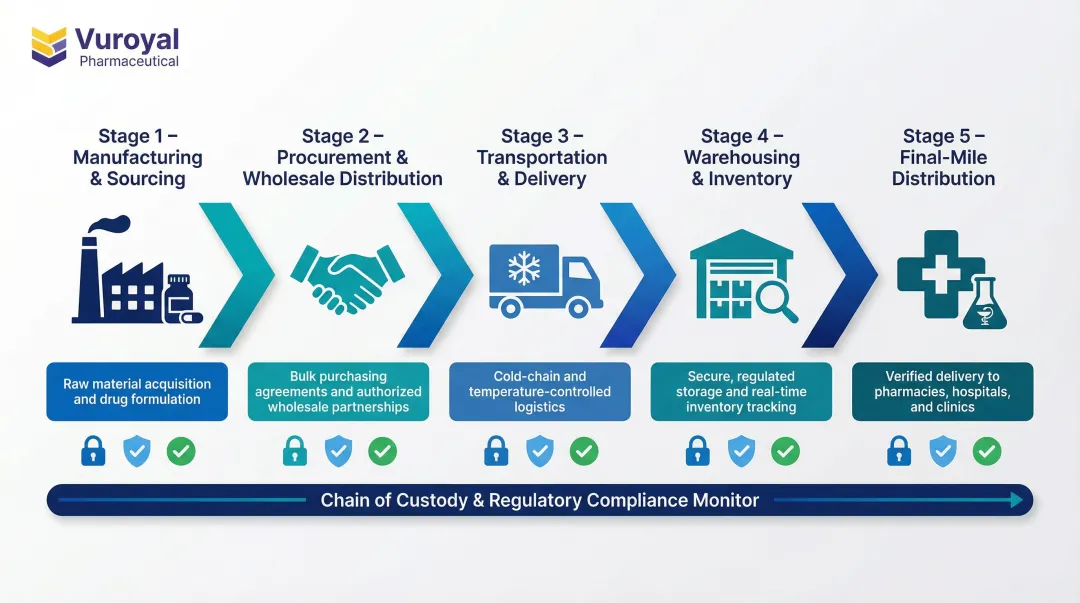
How the Medical Supply Chain Works
The medical supply chain operates as a sequential but interdependent series of stages. A breakdown at any point can compromise the entire chain—and ultimately, the patient or researcher relying on the product. Understanding each stage helps identify where risks concentrate and where specialized expertise adds the most value.
Stage 1: Manufacturing and Sourcing
Pharmaceutical and medical products are produced under controlled conditions at GMP-certified facilities, often with strict temperature and environmental controls. Modern pharmaceutical manufacturing is deeply global: China controls 41% of sole-sourced Key Starting Materials (KSMs) for U.S.-approved APIs, while India supplies roughly 50% of U.S. generic drugs but relies on China for approximately 80% of its own API needs.
This creates fragile, interconnected dependencies. While China accounts for only 8% of direct U.S. API volume, concentration at the KSM level means geopolitical disruptions or quality failures cascade through the entire chain. Single-source reliability matters. When one manufacturer holding large market share closes a facility for quality remediation, shortages of critical drugs like cisplatin and carboplatin follow.
Manufacturing facilities must adhere to Good Manufacturing Practice standards such as FDA cGMP (21 CFR Parts 210 and 211) and EU EudraLex Volume 4. The U.S. and EU operate under a Mutual Recognition Agreement for pharmaceutical GMP inspections, allowing regulators to rely on each other's inspection outcomes.
Stage 2: Procurement and Wholesale Distribution
Licensed pharmaceutical wholesalers and distributors serve as the critical bridge between manufacturers and healthcare end users. This stage involves:
- Verifying supplier credentials and manufacturing documentation
- Confirming product authenticity and regulatory status
- Negotiating pricing and supply agreements
- Sourcing specialty drugs and pharmaceutical excipients internationally
- Managing customs clearance for imported substances
This is where VuRoyal Pharmaceutical operates—as an FDA-registered wholesaler providing one-stop international procurement and customs clearance for specialty drugs and pharmaceutical excipients. For research organizations and clinical trial sponsors, accessing FDA Orange Book Reference Listed Drugs often requires working with specialized distributors who maintain the regulatory credentials and supplier networks to source these products globally.
The procurement stage is particularly complex for specialty substances like vaccine adjuvants (QS-21, Matrix-M) or hard-to-source reference drugs. Distributors must navigate country-specific drug approval requirements, import permits, bilateral trade regulations, and customs documentation while maintaining cold chain integrity.
Stage 3: Transportation and Delivery
Transportation requirements for medical products exceed standard freight specifications:
- Refrigerated trucks and air freight containers must maintain validated temperature ranges
- Biologics, vaccines, and specialty drugs require continuous cold chain (2–8°C for most refrigerated products)
- Every transfer point requires signed chain-of-custody verification
- FDA and DOT regulations govern pharmaceutical transport; DEA requirements apply for controlled substances
The difference between standard and specialty pharmaceutical transportation is significant. Biologics are protein-based and degrade rapidly from temperature excursions, agitation, or UV exposure. A 2019 survey found that 44.6% of respondents reported multiple temperature excursions per year, with 41% of those excursions exceeding four degrees—enough to compromise product integrity.
VuRoyal offers cold chain pharmaceutical transportation services specifically for specialty drugs and research-use substances, ensuring product integrity from procurement through delivery.
Stage 4: Warehousing and Inventory Management
Pharmaceutical warehousing requires specialized infrastructure:
- Temperature-controlled storage zones with continuous environmental monitoring
- Humidity controls for moisture-sensitive products
- Lot tracking systems for recall management and expiration monitoring
- Security protocols for high-value or controlled substances
- FEFO (First Expired, First Out) rotation to prevent expired stock from reaching end users
Poor inventory management leads to stockouts or expired products, both of which disrupt patient care and research timelines. For clinical trials, a single missing reference drug can delay enrollment and regulatory submissions. For hospitals, stockouts force clinicians to substitute alternative medications, increasing medication error risk.
Stage 5: Distribution to End Users
The final mile delivers products to hospitals, clinics, pharmacies, independent research labs, and clinical trial sites. Documentation at point of delivery is critical:
- Temperature integrity proof via data logger records
- Certificates of analysis confirming product specifications
- Chain-of-custody records for every transfer point
- Lot and serial number verification for DSCSA compliance
When distribution fails at this stage, the consequences are immediate: delayed clinical trial enrollment, forced medication substitutions, and gaps in patient care. Every preceding stage exists to make this final handoff reliable.
Key Challenges in Healthcare Logistics
Regulatory Compliance Across Multiple Jurisdictions
Pharmaceutical logistics is regulated by multiple overlapping bodies—the FDA, DEA, state pharmacy boards, and international customs authorities—depending on product type and trade route. Under 21 CFR Part 205, U.S. wholesale distributors must maintain facilities with adequate lighting, ventilation, and security, keep inventories and transaction records for 3 years, and maintain written policies for recalls and outdated drugs.
Compliance failures result in seized shipments, fines, and loss of licensure. Cross-border procurement adds another layer: both the exporting country's regulations and the importing country's drug approval requirements must be met simultaneously.
For example, importing vaccine adjuvant excipients like QS-21 into China requires navigating NMPA registration, while importing into the U.S. requires FDA verification and customs clearance.
Distributors handling controlled substances face additional DEA oversight. Under 21 CFR 1301.74(b), registrants must design and operate systems to disclose suspicious orders—those of unusual size, deviating from normal patterns, or unusual frequency. Failure to maintain effective monitoring systems results in severe penalties: Walgreens paid an $80 million civil penalty in 2013 for failing to report suspicious orders.
Maintaining Cold Chain Integrity
Temperature excursions are one of the most common and costly failure points in pharmaceutical logistics. Even a brief deviation of a few degrees can degrade biologics, vaccines, and other temperature-sensitive drugs, making them ineffective or dangerous.
The global pharmaceutical cold chain market was valued at $384 billion, representing 32% of total pharmaceutical market value but only 0.2% of volume—a value-volume paradox driven by high-cost biologics in immunology and oncology. Cold chain failures remain a persistent problem despite that outsized value.
Maintaining cold chain integrity requires:
- Continuous temperature monitoring through data loggers and IoT-enabled sensors
- Pre-qualified shipping lanes with validated temperature performance
- Proper packaging including insulated containers, phase-change materials, dry ice, and cryogenic systems
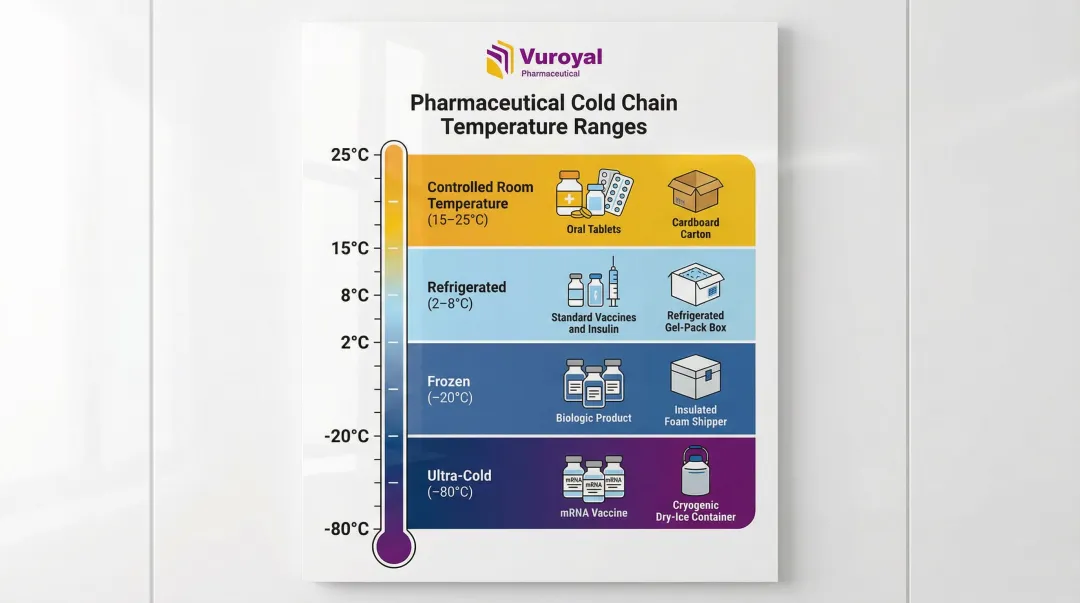
USP <659> defines standard temperature ranges: Refrigerator (2-8°C) and Controlled Room Temperature (20-25°C, with allowable excursions between 15-30°C). For advanced biologics and mRNA-based products, frozen and ultra-cold storage (-20°C to -80°C) may be required.
Counterfeit Drugs and Supply Chain Security
Counterfeit and substandard products pose a serious threat across global pharmaceutical supply chains. The World Health Organization estimates that at least 1 in 10 medical products in low- and middle-income countries is substandard or falsified, costing an estimated $30.5 billion annually.
Interpol's Operation Pangea XVII (2025) seized $65 million in illicit pharmaceuticals, leading to 769 arrests across 90 countries. In the U.S., the Partnership for Safe Medicines reports that counterfeiters increasingly infiltrate the supply chain with fake Adderall, fentanyl-laced pills, and counterfeit GLP-1s like Ozempic, which have caused hospitalizations for hypoglycemia.
The Drug Supply Chain Security Act (DSCSA) in the U.S. requires serialization and traceability at each step of the supply chain. Working with FDA-registered, licensed distributors with verified sourcing networks is the primary defense against counterfeit risk. VuRoyal holds FDA registration, a Massachusetts-approved pharmaceutical wholesaler license, and employs third-party compliance solutions to ensure adherence to DSCSA requirements.
Supply Disruptions and Demand Forecasting
Healthcare logistics is highly susceptible to supply shocks—drug shortages caused by manufacturer issues, geopolitical events, shipping bottlenecks, or raw material scarcity. As of late 2025, ASHP tracked 216 active drug shortages, with 75% classified as long-standing (started in 2022 or later).
Manufacturing quality issues are the primary cause of shortages. Sterile injectables are the most exposed category: of 102 drugs in active shortage tracked by GAO in July 2024, 71 were sterile injectables. These drugs are complex to manufacture and carry thin margins, leading to underinvestment and market consolidation.
Demand forecasting for pharmaceuticals is operationally challenging, especially for specialty drugs and research-use substances with limited supplier options. Mitigation strategies include:
- Multi-source procurement to avoid single-supplier dependencies
- Safety stock planning for critical medications
- Partnering with distributors who have established supplier networks and exclusive sourcing agreements
VuRoyal's exclusive partnership with Desert King International for vaccine adjuvants provides clients with reliable access to QS-21, Matrix-M, and other specialty excipients through a verified, GMP-certified supply chain.
Cold Chain and Specialty Pharmaceutical Logistics
Cold chain logistics in the pharmaceutical context means an unbroken temperature-controlled supply chain from manufacturing through final delivery. Temperature ranges vary by product type: standard refrigerated (2-8°C), controlled room temperature (15-25°C), frozen (-20°C), and ultra-cold for certain biologics and mRNA-based products (-80°C). Even short-term excursions can disqualify products and must be documented.
Packaging and Technology for Cold Chain Compliance
Specialized packaging solutions used in pharmaceutical cold chain include:
- Insulated shippers with validated thermal performance
- Phase-change materials (PCMs) that maintain target temperatures for extended periods
- Dry ice for frozen shipments
- Cryogenic containers for advanced biologics requiring ultra-cold storage
Temperature monitoring technology provides continuous oversight:
- Data loggers that record temperature throughout transit
- IoT-enabled sensors providing real-time alerts for excursions
- Continuous temperature records required for regulatory submission and GMP compliance
Packaging validation and lane qualification are required for GMP-compliant pharmaceutical shipping. The International Air Transport Association (IATA) developed the CEIV Pharma certification, a "GDP+" program that standardizes handling processes and facility requirements to prevent temperature deviations during air freight.
Specialty Drug Logistics: Vaccines, Biologics, and Research Substances
Specialty pharmaceutical logistics—covering vaccines, adjuvants, biosimilars, and pharmaceutical excipients used in research—demands stricter handling protocols and more detailed regulatory documentation than standard pharmaceuticals. Biologics are protein-based and can degrade rapidly from temperature excursions, agitation, or UV exposure.
Adjuvants represent one of the most handling-sensitive categories within biologics. QS-21, for example, is chemically unstable and prone to spontaneous hydrolysis — its degradation accelerates at warmer temperatures and pH ≥ 7.4. Stability data places it most stable at pH 5.5 in micellar form. Proper storage requires:

- Maintaining temperatures at -20°C
- Avoiding repeated freeze-thaw cycles
- Protecting from UV exposure and agitation throughout transit
VuRoyal provides cold chain transportation services for specialty drugs and research-use substances, covering temperature-controlled storage, regulatory documentation, and customs clearance from procurement through delivery. As the exclusive U.S.-China partner of Desert King International, VuRoyal distributes GMP-certified vaccine adjuvants — including QS-21, Matrix-M, QS-7, QS-17, and QS-18 — for human vaccine programs, with full cold chain oversight across international shipments.
Compliance, Regulations, and Certifications in Healthcare Logistics
Regulatory compliance is the foundation of trustworthy healthcare logistics. Every entity touching the pharmaceutical supply chain must hold valid, current licenses and certifications. The compliance structure is multi-level: federal (FDA registration, DEA for controlled substances), state (pharmaceutical wholesaler license), and international (customs registration, country-specific import/export authorization).
Key Certifications and Licenses to Look For
When evaluating a pharmaceutical logistics or distribution partner, verify these essential credentials:
- FDA-registered pharmaceutical wholesaler status — mandatory federal recognition for any entity distributing prescription pharmaceuticals in the U.S.
- State pharmaceutical wholesaler/distributor license — required in every state where distribution occurs; each state sets its own inspection protocols.
- Drug Business Quality Management Standard Certification — confirms adherence to GMP-aligned quality systems for pharmaceutical distribution and storage.
- Customs declaration unit registration — enables legal import/export of controlled substances and specialty drugs across borders.
- DSCSA-compliant serialization and traceability systems — required under the Drug Supply Chain Security Act. The FDA's 1-year stabilization period for electronic tracing ended on November 27, 2024, meaning strict enforcement for electronic interoperability is now in effect. Small dispensers (pharmacies with ≤25 full-time staff) have an exemption until November 27, 2026.

VuRoyal holds FDA registration, a Massachusetts-approved pharmaceutical wholesaler license, a drug business license, and a drug business quality management standard certification. Third-party compliance tools monitor serialization data and flag traceability gaps in real time, keeping VuRoyal's operations aligned with both state licensing requirements and DSCSA mandates.
International Compliance Considerations
Cross-border pharmaceutical procurement adds additional compliance layers: country-specific drug approval requirements, import permits, customs documentation, and cross-border trade rules.
Importers of specialty pharmaceutical substances — excipients like QS-21 or reference listed drugs — need distributors experienced in international regulatory filing and customs clearance, not just domestic shipping providers.
Regulatory requirements vary significantly by jurisdiction. Two key markets illustrate this complexity:
- United States: Excipients like QS-21 are not approved as standalone products. They are reviewed within a New Drug Application (NDA) or Biologics License Application (BLA). The FDA's Inactive Ingredient Database (IID) helps sponsors justify safety during review.
- China: The National Medical Products Administration (NMPA) is tightening excipient oversight. Effective January 1, 2026, the NMPA's Pharmaceutical Excipients Annex requires manufacturers to establish change management systems and conduct regular quality assessments of raw material suppliers.
VuRoyal successfully completed the agency application for registration of pharmaceutical excipient Quillaja saponin QS-21 with China's NMPA in September 2025, with the product entering Public Notice Phase I status. This milestone marks QS-21's entry into China's formal regulatory pathway for domestic compliant application.
Frequently Asked Questions
What is healthcare logistics?
Healthcare logistics is the specialized management of the pharmaceutical and medical supply chain—encompassing procurement, transportation, cold chain management, storage, and distribution—ensuring the right medical products reach the right patients and healthcare facilities on time and in compliant condition.
What certifications are important in healthcare logistics?
Core credentials include FDA registration, state pharmaceutical wholesaler licenses, drug business quality management certifications, and DSCSA compliance for traceability. International operations also require customs registration and country-specific import authorization.
What is cold chain logistics in healthcare?
Cold chain logistics manages temperature-sensitive pharmaceutical products—such as vaccines, biologics, and specialty drugs—through an unbroken temperature-controlled process from manufacturing to delivery, using specialized packaging, refrigerated transport, and continuous temperature monitoring to prevent product degradation.
How does healthcare logistics differ from regular logistics?
Healthcare logistics requires strict regulatory oversight, mandatory cold chain controls, and zero tolerance for delivery failures given the direct patient safety implications. Chain-of-custody documentation requirements also apply at every step—obligations that standard commercial freight does not face.
What are the main stages of the pharmaceutical supply chain?
The five stages are:
- Manufacturing and sourcing
- Procurement and wholesale distribution
- Transportation
- Warehousing and inventory management
- Final-mile distribution to healthcare providers or research organizations
Compliance and product integrity must be maintained at every stage.
What role does a pharmaceutical wholesaler play in healthcare logistics?
Licensed pharmaceutical wholesalers act as the verified intermediary between drug manufacturers and healthcare end users. They handle sourcing, authenticity verification, regulatory documentation, customs clearance, and reliable delivery—making them essential partners for hospitals, pharmacies, and research organizations that cannot procure directly from manufacturers.


