
Introduction to Pharmaceutical Warehousing
Pharmaceutical supply chains operate under immense pressure. Unlike standard storage facilities, pharmaceutical warehouses serve as the last line of defense between a drug's integrity and patient safety. Temperature excursions cost the industry an estimated $35 billion annually, with the World Health Organization reporting that up to 50% of global vaccines are wasted due to cold chain failures. Even minor storage lapses can render life-saving medications ineffective or dangerous — with direct consequences for patient outcomes.
This guide covers the regulatory standards that govern pharmaceutical warehousing—including GMP, GDP, and GWP frameworks—and explores storage requirements, inventory best practices, and optimization strategies. Whether you're managing a pharmaceutical distribution facility or evaluating a 3PL partner, these fundamentals determine whether your supply chain holds up under regulatory scrutiny.
TLDR — Key Takeaways
- GMP, GDP, and GWP compliance — enforced by the FDA and EMA — is mandatory for all pharmaceutical warehouses
- Temperature control failures drive $35B in annual losses; IoT monitoring and validated mapping are essential safeguards
- FEFO inventory rotation, batch tracking, and WMS integration ensure product integrity and recall readiness
- DSCSA requires 2D barcode serialization for unit-level tracing, with full compliance deadlines now in effect
- Working with an FDA-registered distributor like VuRoyal Pharmaceutical provides built-in compliance infrastructure for sourcing RLDs and vaccine adjuvants
What Is Pharmaceutical Warehousing?
Pharmaceutical warehousing covers the storage, handling, and distribution of pharmaceutical products—prescription drugs, biologics, vaccines, and medical compounds—within controlled environments designed to preserve product safety and efficacy. Unlike general warehousing, these facilities operate under strict regulatory oversight from agencies like the FDA and EMA, with documented procedures governing every aspect of operations.
That regulatory framework underpins the supply chain role these facilities play. They receive products from manufacturers, hold them under validated conditions, and distribute to healthcare providers, pharmacies, clinical trial sites, and research organizations. Throughout, the warehouse must ensure identity, strength, quality, and purity remain unaffected—as required by FDA 21 CFR 211.142.
Types of Pharmaceutical Warehouses
Pharmaceutical storage facilities are purpose-built or adapted to maintain specific environmental conditions based on product requirements:
| Warehouse Type | Temperature Range | Typical Products | Key Standard |
|---|---|---|---|
| Ambient / General | 20–25°C (excursions: 15–30°C) | Oral solids, stable pharmaceuticals | USP <659> |
| Refrigerated / Cold Chain | 2–8°C | Vaccines, insulin, biologics | WHO Annex 9 |
| Frozen Storage | -25°C to -10°C (or -60°C to -80°C for ultra-sensitive biologics) | Plasma, mRNA vaccines | WHO Annex 9 |
| Controlled Substance | Ambient with enhanced security | Narcotics, Schedule-class drugs | DEA 21 CFR 1301.72 |
| Clinical Trial / IMP | Product-specific | Investigational Medicinal Products | EudraLex Vol. 4, Annex 13 |
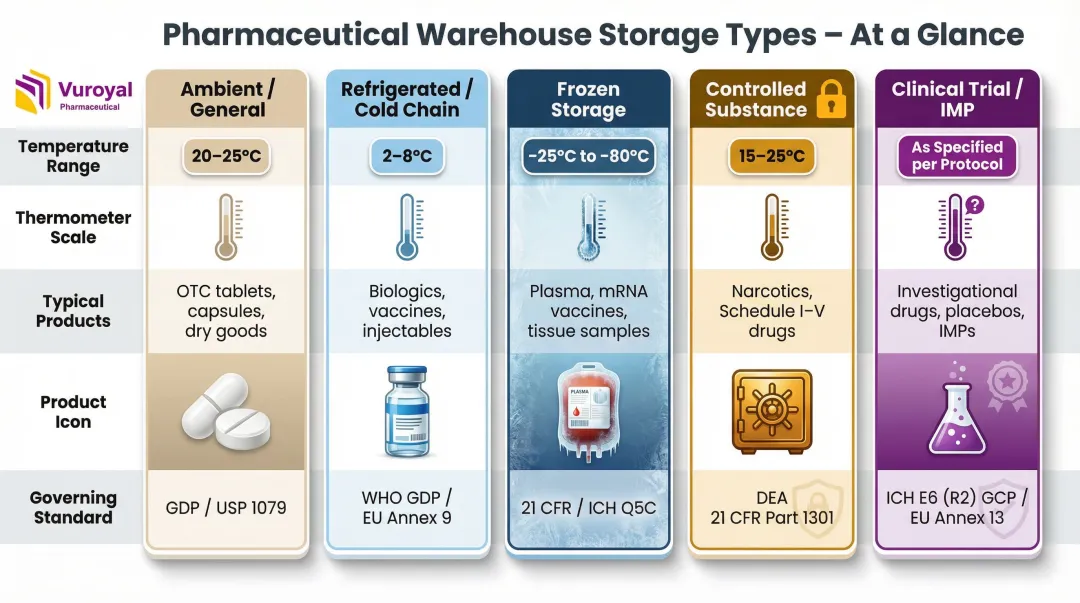
Each type carries distinct infrastructure and compliance requirements. Cold chain and frozen facilities, for example, require continuous environmental monitoring—sensors accurate to ±0.5°C logging conditions at least six times per hour. Controlled substance warehouses add vaults, steel cabinets, and electronic alarm systems on top of standard pharmaceutical controls. Clinical trial facilities go further still, managing blinding integrity and randomization code protocols that don't apply in commercial distribution.
Regulatory Framework: GMP, GDP, GWP & FDA Requirements
Pharmaceutical warehouses operate within a complex regulatory framework designed to protect product integrity from manufacturer to patient.
Good Manufacturing Practice (GMP)
GMP establishes baseline standards for how pharmaceutical products are produced and stored. Under FDA 21 CFR Part 211, requirements cover equipment qualification, personnel training, documentation protocols, and environmental controls. Facilities must maintain appropriate conditions of temperature, humidity, and light to prevent degradation.
The FDA and EMA conduct regular inspections to verify GMP compliance — making documented procedures a non-negotiable operational requirement.
Good Distribution Practice (GDP)
GDP governs how medicines are stored and transported throughout the supply chain. The EMA's 2013/C 343/01 guidelines require wholesale distributors to maintain a quality system, designate a Responsible Person, and ensure products stay within acceptable temperature limits during transit. GDP mandates documentation integrity and prevention of contamination or tampering from manufacturer to end-user.
Good Warehousing Practice (GWP)
Where GDP addresses the full distribution chain, GWP focuses specifically on the storage environment itself. Though incorporated within GMP and GDP frameworks, GWP addresses facility-specific concerns: temperature control protocols, pest management, product segregation, security measures, and staff qualification requirements.
FDA Registration and DSCSA Compliance
FDA registration is mandatory for pharmaceutical wholesalers and distributors operating in the U.S. The Drug Supply Chain Security Act (DSCSA) adds a further layer, requiring electronic traceability of prescription drugs at the unit level to prevent counterfeit products from entering the supply chain.
Key DSCSA requirements currently in effect:
- The 1-year stabilization period for package-level electronic tracing ended November 27, 2024
- Small dispenser exemptions extend to November 27, 2026
- All trading partners must exchange transaction information electronically using 2D data matrix barcodes containing the NDC, serial number, lot number, and expiration date
Consequences of Non-Compliance
Regulatory violations trigger severe penalties. The FDA issued a warning letter to MD Pharmaceutical Supply for failing to monitor API storage conditions—temperature probes were non-operational and humidity wasn't monitored at all. Sterling Distributors received citations for engaging in wholesale distribution without appropriate state licensure and failing to quarantine suspect counterfeit products.
Consequences include:
- Product recalls affecting entire distribution networks
- FDA warning letters and consent decrees
- License revocation and facility closure
- Financial penalties and legal liability
- Reputational damage and loss of customer trust

For distributors handling specialty products — such as reference listed drugs or vaccine adjuvants — audit trails and validated SOPs aren't just inspection requirements. They're the operational backbone that keeps product integrity intact from the point of receipt through final delivery.
Temperature Control & Controlled Storage Conditions
Temperature control is the cornerstone of pharmaceutical warehousing. Products stored outside specified ranges—even briefly—can undergo irreversible degradation that renders them ineffective or unsafe.
Standard Pharmaceutical Temperature Zones
USP <659> defines authoritative temperature ranges for pharmaceutical storage:
| Zone | Range | Key Products |
|---|---|---|
| Controlled Room Temperature (CRT) | 20–25°C; excursions 15–30°C permitted; transient spikes to 40°C allowed <24 hrs if mean kinetic temperature ≤25°C | Oral solid dosage forms |
| Refrigerated | 2–8°C; even brief excursions above 8°C can compromise potency | Vaccines, insulin, biologics |
| Frozen | -25°C to -10°C; some articles require below -20°C (±10°C) | Plasma, biological materials |
| Ultra-Cold | -60°C to -80°C | mRNA vaccines |
Temperature Mapping and Validation
Temperature mapping is a validated survey of a storage space to identify hot spots and cold zones. WHO TRS 961 Annex 9 and USP <1079> require studies lasting at least two weeks to capture workflow variations, including weekends. Facilities must also conduct seasonal mapping in both summer and winter to document temperature distribution under anticipated environmental extremes.
Those findings directly shape storage layout, HVAC placement, and monitoring sensor locations. High-value or sensitive products should never be placed in identified problem zones without additional controls.
Continuous Environmental Monitoring
WHO Annex 9 mandates continuous monitoring systems for temperature-controlled rooms with specific requirements:
- Sensor accuracy: ±0.5°C or better
- Logging frequency: Minimum six times per hour
- Calibration: Against certified, traceable reference standards at least annually
- Alarm verification: Checked at least every six months
- Humidity monitoring: ±5% RH accuracy for products adversely affected by moisture

Systems must generate automated alerts when conditions deviate from acceptable ranges, enabling immediate corrective action. Electronic records used to meet regulatory requirements must comply with FDA 21 CFR Part 11, including secure, time-stamped audit trails and restricted system access.
Humidity and Air Quality Control
Excess moisture degrades tablets and compromises packaging integrity. Particulate contamination in storage areas near cleanrooms can compromise sterile products. To address both risks, facilities typically deploy:
- Dehumidifiers to maintain target RH levels
- HEPA filtration to remove airborne particulates
- Positive-pressure environments to prevent contaminated air ingress
Cold Chain Beyond the Warehouse
Cold chain management extends through transportation and last-mile delivery. VuRoyal Pharmaceutical provides cold chain transportation as part of its specialty drug logistics services, maintaining documented temperature control for products like vaccine adjuvants (QS-21, Matrix-M™) and biologics from warehouse release through final destination.
Pharmaceutical Inventory Management Best Practices
Advanced inventory management protects product integrity and ensures regulatory compliance through the entire storage lifecycle.
FEFO: The Pharmaceutical Standard
While FDA 21 CFR 211.150 requires distributing the "oldest approved stock first" (FIFO), global GDP guidelines—including WHO Annex 9 and EMA 2013/C 343/01—strictly mandate First-Expired, First-Out (FEFO) for pharmaceutical products. FEFO prioritizes distribution based on expiration date rather than receipt date, minimizing waste and ensuring patients receive products with adequate remaining shelf life.
Warehouse Management Systems must be configured to automatically route FEFO picks, preventing the distribution of short-dated products when longer-dated inventory is available.
Supply Chain Traceability Identifiers
Effective FEFO enforcement depends on accurate data capture. Pharmaceutical warehouses must track multiple identifiers for each product:
- Batch/lot numbers for recall traceability
- Expiration dates to drive FEFO routing decisions
- Unique Identifying Numbers (UIN) for unit-level tracking under DSCSA
- NDC codes to prevent mix-ups across multi-product inventories
- Serial numbers enabling package-level traceability from manufacturer to dispenser

Barcoding and Serialization
The DSCSA requires 2D data matrix barcodes on each package and homogeneous case. These barcodes encode the NDC, a unique alphanumeric serial number (up to 20 characters), lot number, and expiration date. The FDA recommends human-readable formatting as: NDC: [insert NDC], SERIAL: [insert serial number], LOT: [insert lot number], EXP: [insert expiration date].
RFID tags and automated scanning at each handling stage—receiving, put-away, picking, and shipping—reduce human error and maintain an unbroken traceability record from manufacturer to dispenser.
Physical and Electronic Product Segregation
FDA 21 CFR 211.80 and 211.142 require that each lot be clearly labeled by status: quarantined, approved, or rejected. Facilities must:
- Physically separate quarantined, approved, rejected, and recalled products
- Use designated areas or locked zones for non-conforming inventory
- Mirror physical segregation in the WMS with electronic status labels (Hold, Released, Approved, Rejected, Quarantined)
- Prevent accidental distribution of non-conforming products through system controls
Receiving and Quality Inspection Workflows
Receiving is where product integrity is confirmed or compromised. Every inbound shipment requires:
- Verifying shipment documentation against purchase orders
- Inspecting for temperature excursions during transit using data loggers
- Checking packaging integrity for signs of damage or tampering
- Logging batch data and expiration dates prior to release into general stock
- Quarantining products pending quality control unit approval
Technology Tools for Pharmaceutical Warehouse Optimization
Three technology layers — warehouse management systems, IoT environmental monitoring, and blockchain traceability — work together to keep pharmaceutical warehouses accurate, compliant, and audit-ready. Each addresses a distinct gap that manual processes can't reliably close.
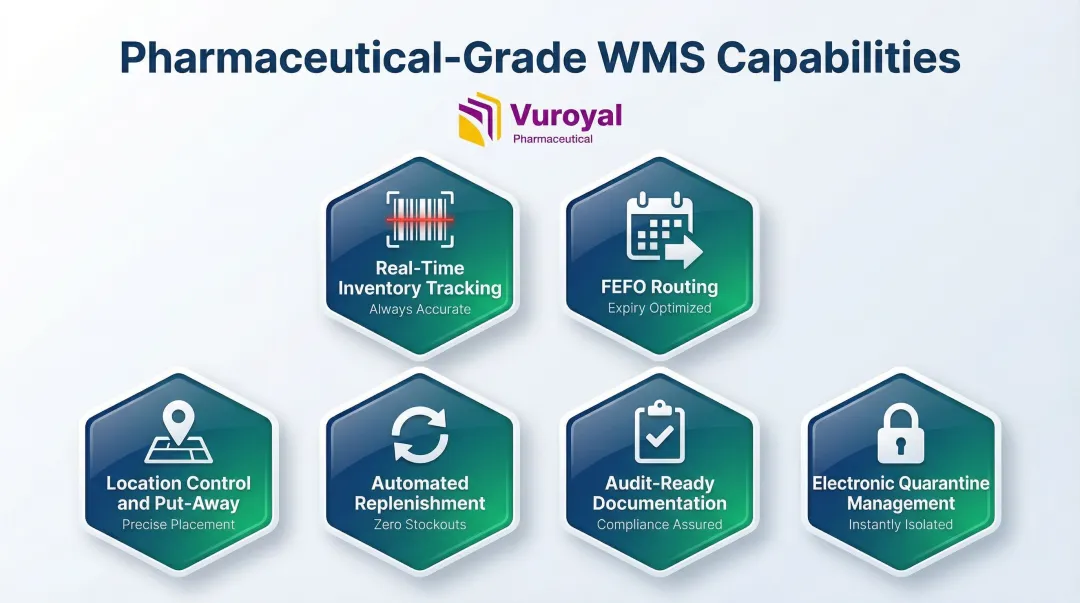
Warehouse Management Systems (WMS)
A pharmaceutical-grade WMS provides:
- Real-time inventory tracking with lot and serial number control
- Automated expiry date management and FEFO routing
- Location control and directed put-away/picking
- Automated replenishment triggers based on min/max levels
- Audit-ready documentation for regulatory inspections
- Electronic quarantine status management
One study on Lean Warehousing with RFID-based WMS found picking accuracy reached 99.5%, with measurable reductions in storage cycle times — a compelling case for integrated tracking over manual methods.
IoT Sensors and Real-Time Monitoring
WMS platforms become significantly more powerful when paired with live environmental data. IoT-connected sensors create continuous logs for temperature, humidity, and security events — and when integrated with WMS, they enable:
- Automated flagging of non-conforming conditions
- Real-time alerts to designated personnel
- Support for deviation investigations with time-stamped records
- Compliance with 21 CFR Part 11 for electronic records
Under 21 CFR Part 11, your system must generate secure, computer-generated audit trails and enforce access controls that restrict actions to authorized personnel only.
Blockchain for Supply Chain Integrity
The FDA's MediLedger pilot demonstrated that blockchain can manage DSCSA interoperability, routing, and verification without exposing proprietary trading partner data. That's a meaningful proof point for an industry where supply chain visibility and confidentiality must coexist.
In practice, blockchain creates immutable records of product movement from manufacturer to patient. For pharmaceutical distributors, this translates to faster recall execution, reduced counterfeiting risk through decentralized verification, and a defensible audit trail across multi-party distribution networks — all in a single system of record.
Security, Staff Training & Recall Management
Protecting pharmaceutical inventory requires physical security, qualified personnel, and rapid-response recall protocols.
Physical Security Requirements
DEA 21 CFR 1301.72 mandates strict physical security for controlled substances. Schedule III-V substances must be stored in:
- A safe or steel cabinet
- A vault equipped with electronic alarm systems transmitting signals directly to central station protection agencies or local police
- A secure cage with ceiling constructed of No. 10 gauge steel fabric
All pharmaceutical warehouses should implement:
- Restricted access controls (key card, biometric)
- 24/7 CCTV surveillance
- Alarmed entry points
- Dedicated secure areas for high-value drugs
Security failures can result in product theft, diversion, and counterfeiting. Under 21 CFR 1301.74, DEA-registered facilities must notify the DEA Field Division Office within one business day of discovering theft or significant loss, then submit DEA Form 106 within 45 calendar days.
Security controls are only as effective as the people operating them — which makes staff training equally critical.
Staff Training Requirements
FDA 21 CFR 211.25 requires that each person engaged in holding drug products have appropriate education, training, and experience. WHO Annex 9 reinforces this with requirements for regular and systematic training covering:
- Proper handling of pharmaceutical products
- Temperature excursion response procedures
- Security protocols and access controls
- Documentation standards and GMP principles
- Specialized training for controlled, hazardous, or temperature-sensitive products
Training records must be maintained within the quality management system — typically with employee sign-off logs and completion dates — and made available during regulatory inspections. A documented training program also supports recall readiness, since personnel who understand traceability procedures can execute product pulls faster.
Recall Readiness and Expired Product Management
FDA 21 CFR 7.50 classifies recalls into three categories based on health risk. Class I recalls — involving a serious probability of adverse health consequences or death — require immediate action and leave no margin for slow response.
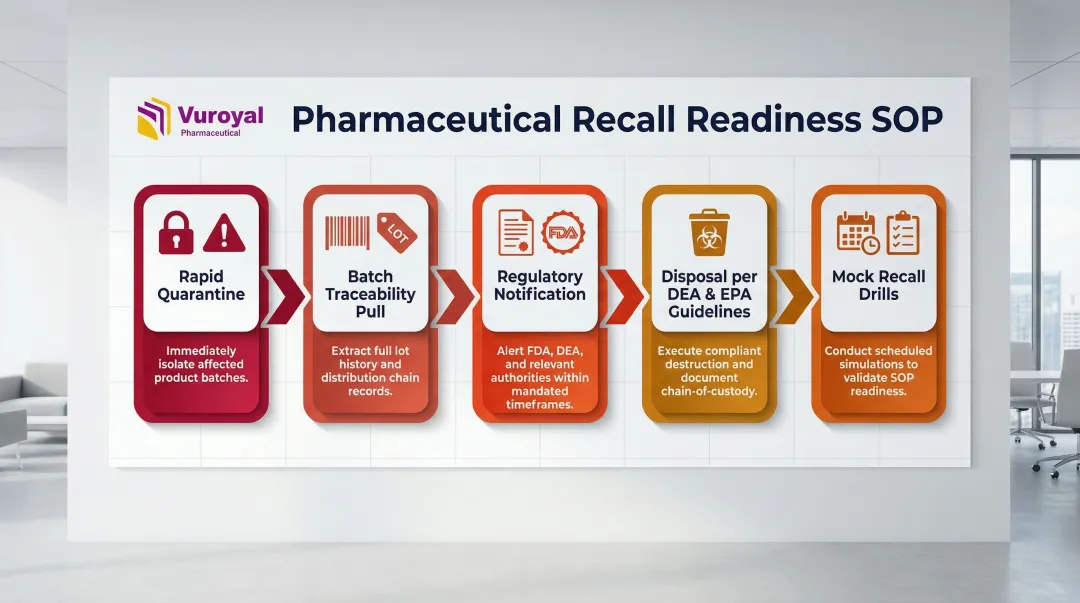
Pharmaceutical warehouses must maintain documented SOPs for:
- Rapid product quarantine upon recall notification
- Batch traceability pull using lot numbers and serialization data
- Regulatory notification to FDA and affected customers
- Disposal of expired or recalled products following DEA and EPA guidelines
- Mock recall drills conducted periodically to validate response time and accuracy
Under 21 CFR 211.150, distributors must maintain a system by which the distribution of each lot can be readily determined to facilitate recall.
Frequently Asked Questions
What are the 5 types of warehouses and their functions?
The five pharmaceutical warehouse types are:
- Ambient/general storage (CRT 15-25°C) for oral solids and stable compounds
- Refrigerated cold chain (2-8°C) for vaccines and biologics
- Frozen storage (-25°C to -10°C) for plasma and sensitive compounds
- Controlled substance warehouses with enhanced security for narcotics
- Specialized warehouses for clinical trial materials or niche compounds requiring segregated storage
What is the difference between GMP and GDP in pharmaceutical warehousing?
GMP (Good Manufacturing Practice) governs production and storage quality standards, covering equipment, personnel, and environmental controls. GDP (Good Distribution Practice) governs the handling, transportation, and distribution of finished pharmaceutical products throughout the supply chain. Compliant warehousing operations must satisfy both frameworks.
What temperature is required for pharmaceutical storage?
Requirements vary by product: Controlled Room Temperature (15-25°C) covers most oral medications, Refrigerated (2-8°C) applies to vaccines and biologics, and Frozen (-20°C or below) is required for plasma and certain compounds. Always confirm against specific product labeling and USP <659> standards.
What is Good Warehousing Practice (GWP)?
GWP is the set of standards governing how pharmaceutical products are received, stored, and handled within a warehouse. It covers environmental controls, temperature and humidity monitoring, pest management, product segregation, security measures, and staff qualifications—generally subsumed within broader GMP and GDP regulatory frameworks.
How do pharmaceutical warehouses handle product recalls?
Warehouses use batch-level traceability—lot numbers and serialization—to rapidly identify, quarantine, and document affected products while notifying regulators and customers within required timeframes. Facilities run mock recall drills regularly to verify complete traceability before a real event occurs.
What licenses are required to operate a pharmaceutical warehouse or distribute pharmaceutical products?
Requirements vary by jurisdiction but typically include FDA registration, state-level pharmaceutical wholesaler licenses, drug business licenses, and quality management certifications. VuRoyal Pharmaceutical, for instance, holds FDA registration and a Massachusetts-approved wholesaler license, covering the full scope of compliant pharmaceutical distribution.


