
Introduction
Clinical supply chains for temperature-sensitive drugs operate under far higher stakes than standard pharmaceutical logistics. A temperature excursion doesn't just waste product — it can invalidate a clinical trial, erase years of research, and put patients at risk. The pharmaceutical industry loses approximately $35 billion annually due to failures in temperature-controlled logistics, spanning lost product, trial disruptions, and supply chain inefficiencies.
The scale of waste is striking. The World Health Organization estimates that up to 50% of vaccines are wasted globally every year, largely due to inadequate temperature control and broken cold-chain logistics.
This guide gives clinical trial sponsors and research organizations a practical reference covering cold chain definitions, temperature categories, core components, key challenges, and best practices. With biologics now representing 49% of the global pharmaceutical pipeline, cold chain integrity is no longer optional — it determines whether a trial succeeds or fails.
TLDR:
- Cold chain failures cost the pharmaceutical industry $35 billion annually and waste 50% of vaccines globally
- Biologics comprise 49% of the pipeline, requiring refrigerated (2-8°C), frozen (-20 to -80°C), or cryogenic (<-150°C) conditions
- Three core components: qualified cold storage, validated transport/packaging, and continuous monitoring with audit-ready documentation — each essential to regulatory compliance
- Real-time IoT monitoring reduces failure rates from 2-5% to <0.3% compared to passive data loggers
- FDA, EMA, and NMPA each impose distinct requirements, making specialized import expertise essential for multi-jurisdiction trials
What Is Cold Chain Clinical Supply Management?
Cold chain clinical supply management is the specialized discipline of maintaining unbroken, validated temperature-controlled conditions for investigational medicinal products (IMPs), biologics, vaccines, and other temperature-sensitive clinical materials — from manufacturing through storage, transit, site delivery, and final patient administration.
Unlike general pharmaceutical cold chain logistics, clinical supply involves added complexity:
- Small, unpredictable batch sizes that require flexible manufacturing and distribution
- Multi-site global trial distribution spanning dozens or hundreds of investigational sites across different countries
- Blinding requirements that complicate labeling and inventory management
- Strict regulatory scrutiny under ICH, FDA, and EMA guidelines for clinical materials — requirements not typically present in commercial supply chains
The product categories requiring cold chain management now span biologics (monoclonal antibodies, cell and gene therapies), mRNA vaccines, vaccine adjuvants, oncology drugs, and insulin — each demanding precise, uninterrupted temperature control throughout the supply chain.
The market scale reflects this reality. Biologics currently represent 49% of the global pharmaceutical pipeline, and cold chain medicines accounted for $384 billion — or 32% — of total pharmaceutical market value in 2021, excluding vaccines and COVID-19 therapeutics.
Why Cold Chain Integrity Is Non-Negotiable in Clinical Trials
Product Integrity Risk: Irreversible Molecular Damage
Even brief temperature excursions outside the validated range can irreversibly alter a biologic's molecular structure. According to ICH Q5C guidelines, biotechnological and biological products are highly sensitive to environmental factors such as temperature changes, oxidation, and light. Exposure outside recommended ranges disrupts noncovalent and covalent forces, leading to:
- Protein aggregation and denaturation
- Ice crystal formation in frozen products
- Loss of biological activity
- Generation of toxic degradation products
Once these molecular changes occur, the IMP is rendered ineffective or potentially harmful before it ever reaches a trial participant — and the damage cannot be reversed.
Patient Safety Consequences
Those molecular changes don't stay contained to the vial — they translate directly into risk for trial participants. Administering a temperature-compromised IMP can result in:
- Toxic degradation products forming when proteins denature, triggering adverse events
- Loss of therapeutic effect, leaving participants exposed to trial risks with no clinical benefit
- False safety or efficacy signals that undermine the validity of the entire trial
When compromised product reaches a participant, the harm is twofold: individual welfare is put at risk, and the scientific record is corrupted.
Trial Integrity and Data Consequences
If a cold chain failure goes undetected and compromised product is administered, trial efficacy data becomes unreliable. This can lead to:
- False negatives that incorrectly suggest a promising therapy doesn't work
- Stalled or terminated development programs built on invalid data
- Years of wasted research and millions in lost investment
- Delayed access to therapies for the patients who need them most
Financial and Regulatory Consequences
The financial impact of cold chain excursions extends far beyond the physical product:
| Cost Category | Financial Impact | Context |
|---|---|---|
| IMP Replacement | $100,000 - $500,000 per shipment | Manufacturing, testing, and shipping costs for a failed batch |
| Trial Delays | $500,000 per day | Average cost including direct expenses and lost commercialization opportunity |
| Opportunity Cost | $200,000 - $1,000,000 | Cost of a 30-day delay from replacement manufacturing |

Regulatory agencies require documented evidence of temperature control throughout the supply chain. Failures can trigger audits, warning letters, or clinical holds that halt trial progress entirely.
Unique Vulnerability of Advanced Therapies
Several emerging therapy categories have stability windows measured in hours — not days — when exposed to temperature excursions:
- Cell and gene therapies: typically stored at -80°C or cryogenic conditions; highly sensitive to freeze-thaw cycles
- mRNA constructs: require ultra-cold storage (-60°C to -80°C); degradation can begin within minutes at ambient temperature
- Novel vaccine adjuvants (such as QS-21): require controlled cold chain from manufacture through point of use, with narrow validated ranges
For these products, the margin for error isn't just narrow — in many cases, a single undetected excursion is enough to render an entire lot unusable.
Temperature Ranges and Product Categories in Clinical Supply
Clinical IMPs fall into three distinct temperature bands, each requiring different validated packaging systems, qualified carriers, storage equipment, and monitoring protocols.
Standard Temperature Bands
| Temperature Band | Range | Clinical Product Examples |
|---|---|---|
| Refrigerated | 2°C to 8°C | Most vaccines, monoclonal antibodies (e.g., Trastuzumab/Herceptin: "Store reconstituted Herceptin at 2-8°C"), insulin |
| Frozen / Ultra-Cold | -20°C to -80°C | Biologics, advanced therapy products, mRNA vaccines (e.g., Comirnaty: "Store single dose frozen vials... in a freezer at -90°C to -60°C") |
| Cryogenic | ≤ -120°C to -150°C | CAR-T cell therapies (e.g., Tisagenlecleucel/Kymriah: "Store infusion bag(s) in a temperature-monitored system less than or equal to minus 120°C, e.g., in the vapor phase of liquid nitrogen"), viral vectors, gene therapy products |

Each temperature band requires entirely different infrastructure. Mismatches between product requirements and logistics capabilities are a leading source of cold chain failures in clinical trials. Each temperature band requires entirely different infrastructure — and mismatches between product requirements and logistics capabilities are a leading source of cold chain failures in clinical trials. That makes knowing your product's exact thermal requirements the first step in building a viable cold chain strategy.
Stability Profiles Drive Strategy
A product's stability profile — determined by stability testing and shelf-life data — sets the acceptable temperature range and defines permissible excursion limits. Without this data in hand before designing the cold chain strategy, you cannot:
- Define acceptable excursion windows
- Select appropriate packaging systems
- Establish quarantine and release criteria
- Respond effectively when deviations occur
The Three Core Components of Cold Chain Management
Cold Storage
Temperature-controlled storage forms the foundation of cold chain integrity. Qualified cold rooms, pharmaceutical-grade refrigerators, ultra-low temperature (ULT) freezers, and cryogenic storage tanks must maintain validated temperature ranges continuously, even during power fluctuations or door openings.
Qualification and Validation Requirements:
Facilities used for clinical IMP storage must undergo rigorous qualification under GDP and GMP guidelines:
- Thermal mapping (temperature distribution assessment) to identify hot and cold spots within storage areas
- Equipment qualification (IQ/OQ/PQ) to demonstrate that conditions are consistently maintained
- Calibration at defined intervals traceable to national or international standards
- Documented protocols for all qualification activities
EU GDP requires an initial temperature mapping exercise on storage areas before use, under representative conditions, to locate monitoring equipment in areas that experience extreme fluctuations.
Backup and Redundancy Systems:
Storage failures represent catastrophic risk. Essential backup systems include:
- Alarm systems for temperature deviations with 24/7 monitoring
- Redundancy mechanisms (backup power, secondary cooling systems)
- Emergency response SOPs to protect product if primary storage fails
- Documented deviation and corrective action procedures

A study of medication refrigerators found that 44% reached temperatures >8°C, confirming that household-grade refrigerators used in clinical settings are unfit for pharmaceutical storage.
Cold Transport and Packaging
Validated temperature-controlled packaging systems fall into two primary categories:
Passive Packaging:
- Insulated shippers with phase-change materials or dry ice
- Finite amount of pre-conditioned coolant without thermostatic regulation
- Cost: $400 - $1,200 per shipment
- Best for: Short transit (<72 hours), established lanes, frozen products
Active Containers:
- Battery-powered refrigerated units with thermostatic regulation
- Continuous temperature control using electricity or fuel sources
- Cost: $2,000 - $5,000 monthly rental
- Best for: Ultra-cold shipments, long transit (5-7 days), remote sites
Cryogenic Dry-Shippers:
- Specialized containers using liquid nitrogen for ultracold transport
- Required for cell therapies and gene therapy products requiring <-150°C
The Complexity of Clinical Supply Transport:
IMPs often travel across multiple countries, through customs, via air and ground carriers — each handoff is a potential excursion risk. Critical requirements include:
- Pre-validated routing with documented lane qualifications
- Qualified carriers trained to handle the product's temperature requirements
- Packaging validation (testing shipper performance under worst-case temperature conditions for the expected transit duration)
USP <1079> references ISTA 7E and ASTM D4169 for performance testing of shipping containers against shock, vibration, pressure, and thermal transit events. Documentation of this validation is required before any container is used in a clinical trial.
One study evaluating temperature excursions during mail transit found that packages spent an average of 68.3% of their transit time outside the recommended range — underscoring why packaging validation and carrier qualification are non-negotiable.
Cold Chain Monitoring
Temperature Monitoring Devices:
Two primary categories enable visibility into cold chain conditions:
- Passive dataloggers (for example, single-use USB loggers) record temperature at intervals throughout transit, providing post-delivery records for review
- Real-time IoT-connected monitors transmit live temperature and location data, enabling proactive intervention if an excursion begins
That distinction has measurable consequences: standard cold chain logistics using passive data loggers experience 2-5% shipment failure rates, whereas specialized IMP couriers using real-time IoT cellular monitoring achieve <0.3% failure rates.
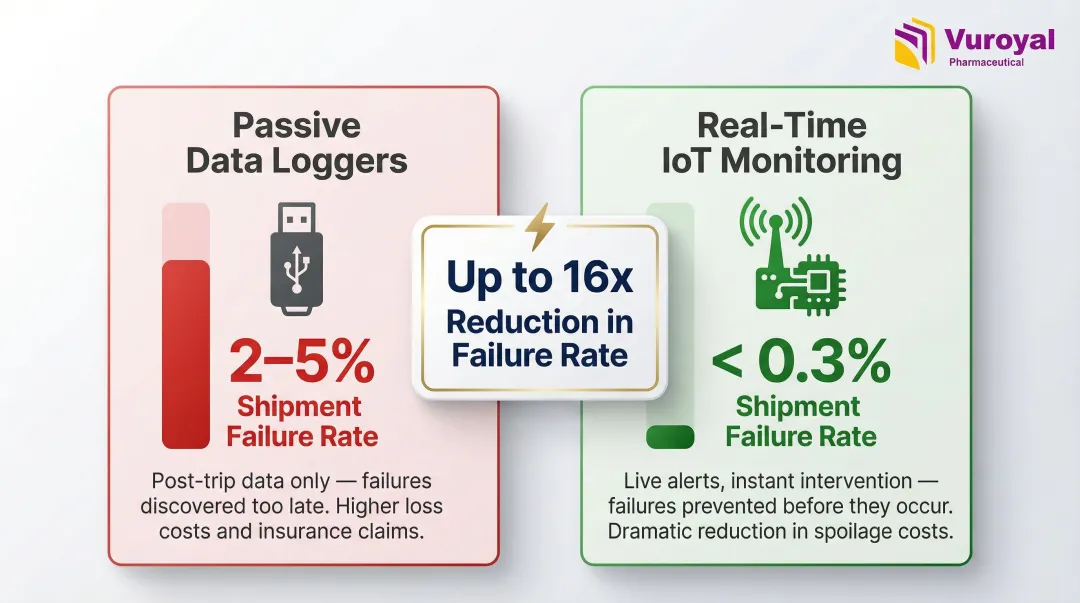
Stationary Monitoring Systems:
For storage facilities, continuous monitoring platforms:
- Record temperature data across all storage units
- Generate automated alarms when deviations occur
- Produce audit-ready compliance reports
- Enable 24/7 oversight without manual checks
Data Integrity and Documentation:
All temperature records must be:
- Traceable with clear chain of custody
- Tamper-proof to prevent unauthorized modifications
- Retained per regulatory guidelines (typically 2-5 years post-trial completion)
- Available for inspection by regulatory authorities
FDA 21 CFR Part 11 requires closed systems to use secure, computer-generated, time-stamped audit trails to independently record the date and time of operator entries and actions that create, modify, or delete electronic records. GDP requires that corrective actions be documented whenever deviations occur.
Key Challenges in Cold Chain Clinical Supply Management
Regulatory Complexity Across Borders
Clinical trials are increasingly global, with ClinicalTrials.gov currently listing 573,913 studies with locations in 225 countries and territories. IMPs must cross multiple regulatory jurisdictions, each with its own cold chain requirements, customs handling rules, and documentation standards.
Regulatory Alignment Gaps:
While major regulatory bodies share similar objectives, distinct procedural differences complicate IMP importation:
- EU/EMA (GDP): Heavy emphasis on quality systems, temperature mapping, and equipment calibration throughout the distribution phase
- US/FDA: Focuses broadly on lifecycle manufacturing, post-market surveillance, and strict 21 CFR 312 import requirements requiring the consignee to be the sponsor, a qualified investigator, or a domestic agent
- China/NMPA: Recent 2025 mandates require overseas marketing authorization holders to legally designate a domestic responsible person to fulfill legal obligations within China; pre-approval commercial-scale batches must comply with GMP, requiring relevant notification of drug inspection results or certificates of GMP compliance from the country of manufacture
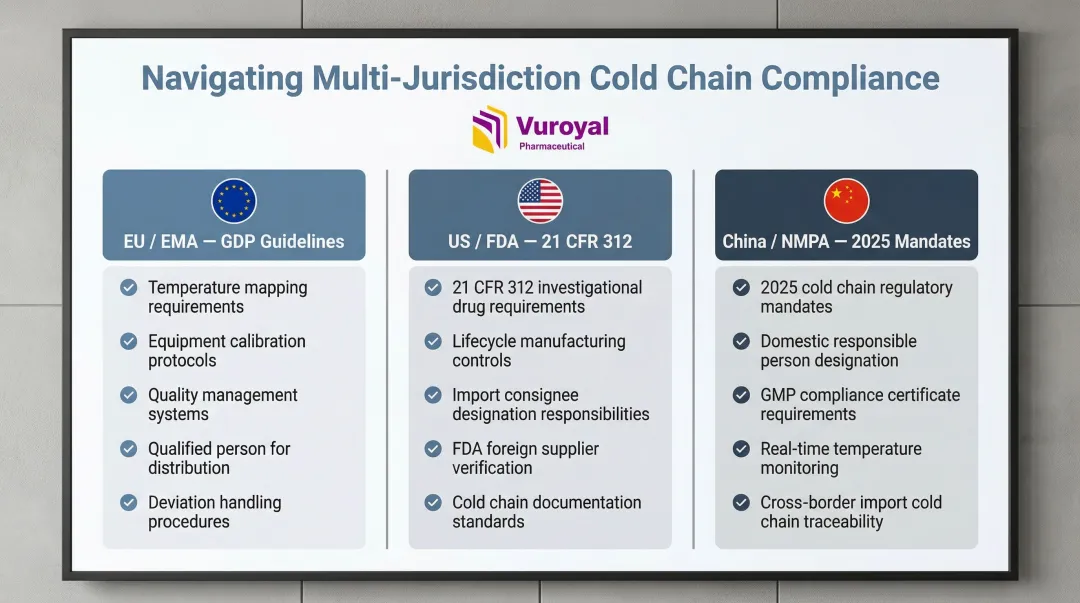
These gaps in cross-border regulatory knowledge frequently cause delays or import failures that can derail trial timelines.
Infrastructure Gaps and Last-Mile Delivery
Maintaining ultracold conditions in remote or underdeveloped clinical site locations — where reliable power, qualified cold storage, and trained staff may be unavailable — is one of the most persistent challenges in clinical trial logistics.
Cell and gene therapy trials face this acutely. A pilot study in Tanzania observed temperature excursions on 82% of transport trips, illustrating the vulnerability of last-mile delivery in low- and middle-income countries (LMICs).
Common last-mile challenges include:
- Unreliable electricity supply at investigational sites
- Inadequate pharmaceutical-grade storage capacity
- Shortages of trained personnel for cold chain management
- Limited access to backup equipment or emergency response capabilities
Temperature Excursion Response and Decision-Making
When an excursion is detected, clinical supply managers must quickly determine whether the product is still within its permissible stability window or must be quarantined and replaced. This requires:
- Excursion response SOPs defined and documented before trial initiation
- Stability data on hand to assess whether the excursion exceeded acceptable limits
- Coordinated response across QA, the sponsor, and the clinical site
- Deviation reports filed for all excursions, regardless of outcome
Without these elements in place, excursion response becomes reactive and inconsistent, increasing the risk of compromised product reaching patients.
Cost and Resource Intensity
Cold chain clinical supply costs more than ambient logistics at every layer — validated infrastructure, qualified personnel, real-time monitoring, and specialized packaging all carry premiums. Active cooling containers, for example, run $2,000–$5,000 per month in rental costs, versus $400–$1,200 for dry ice passive shipping.
Yet those numbers look different when weighed against the alternative. A single day of clinical trial delay costs an average of $500,000 in direct costs and lost opportunity — which means investing in premium logistics upfront is often the lower-cost option.
Managing Complex, Multi-Site Global Distribution
Clinical trials often involve dozens or hundreds of sites across different countries, each receiving small, precisely timed IMP shipments. Coordinating this at scale while maintaining cold chain integrity and meeting each site's delivery windows requires:
- Sophisticated demand planning and forecasting
- Logistics orchestration across multiple carriers and customs authorities
- Site-by-site qualification and training
- Real-time visibility into inventory levels and shipment status
Best Practices for Effective Cold Chain Clinical Supply Management
Develop a Cold Chain Management Plan (CCMP) Early
Clinical trial sponsors should establish a documented CCMP before the trial begins. ICH E6 (Good Clinical Practice) Section 5.13.2 requires the sponsor to determine acceptable storage temperatures, storage conditions, storage times, and reconstitution procedures for the investigational product, and to inform all involved parties (monitors, investigators, pharmacists) of these determinations.
Your CCMP should cover:
- Storage and transport requirements for each temperature band
- Validated packaging specifications with documented performance data
- Monitoring protocols (device types, recording intervals, alarm thresholds)
- Excursion response procedures with decision trees
- Roles and responsibilities across sponsor, CRO, depot, and site personnel
This plan should be reviewed and approved by QA before first IMP shipment.
Invest in Real-Time Visibility and Data-Driven Decision-Making
Real-time temperature and location monitoring platforms that consolidate data from all shipments and storage locations allow supply managers to detect and respond to excursions before product is compromised.
By using real-time monitoring technology, Pfizer achieved less than 0.1% product loss for its COVID-19 vaccines. Automated, onsite monitoring enables swift corrective action in scenarios requiring escalation, such as delays or border issues, reducing supply-chain related product loss and spoilage.
Regularly analyze temperature data to identify recurring risk points in the shipping lane and address them through:
- Lane re-qualification or route changes
- Upgraded packaging systems
- Enhanced carrier training
- Additional monitoring touchpoints
Work with a Qualified, Experienced Cold Chain Logistics Partner
Clinical trial sponsors importing temperature-sensitive materials across international borders face compounding risks at every handoff. Partnering with an FDA-registered distributor experienced in end-to-end cold chain logistics removes much of that exposure.
Look for partners who demonstrate:
- FDA registration or equivalent certification in relevant jurisdictions
- Validated infrastructure covering your required temperature ranges
- Real-time monitoring and documentation with 21 CFR Part 11 compliance
- Regulatory expertise in international customs clearance for pharmaceutical shipments
- Demonstrated experience with clinical trial logistics and IMP handling
VuRoyal Pharmaceutical is an FDA-registered, Massachusetts-approved wholesaler serving research organizations and clinical trial sponsors with exactly this scope. Their end-to-end services cover imported drug approval registration, customs clearance, and cold chain pharmaceutical transportation — with free regulatory guidance included — so temperature-sensitive IMPs reach clinical sites with integrity intact.

Frequently Asked Questions
What is cold supply chain management?
Cold supply chain management is the process of storing, handling, and transporting temperature-sensitive products — including pharmaceuticals, vaccines, and biologics — within defined temperature ranges to preserve their safety, quality, and efficacy from manufacture to end user.
What are the three main components of a cold chain?
The three core components are cold storage, cold transport and packaging, and cold chain monitoring. Cold storage covers temperature-controlled warehousing and pharmaceutical-grade equipment; cold transport includes validated shipping systems and qualified carriers; and cold chain monitoring uses data logging and real-time tracking to maintain visibility and compliance throughout.
What temperature ranges are used in pharmaceutical cold chain management?
Three temperature ranges apply across most pharmaceutical cold chains:
- Refrigerated (2°C to 8°C): most vaccines and biologics
- Frozen (−20°C to −80°C): advanced therapy products
- Cryogenic (below −150°C): cell and gene therapies, using liquid nitrogen
What regulatory requirements apply to cold chain management in clinical trials?
Clinical trial cold chains must comply with GDP and GMP guidelines, FDA 21 CFR regulations, and EMA guidelines. In practice, this means validated storage and transport systems, continuous temperature monitoring, audit-ready documentation, and qualified personnel.
What happens when a cold chain excursion occurs during a clinical trial?
The affected product must be quarantined immediately, the excursion assessed against the product's stability data to determine if it remains within acceptable limits, and a documented deviation report filed. A replacement shipment may be required if the product is deemed compromised.
How do you choose a cold chain logistics partner for clinical supply?
Prioritize partners who meet these criteria:
- FDA-registered or equivalently certified in their operating region
- Validated infrastructure covering your required temperature ranges
- Real-time monitoring with audit-ready documentation
- Demonstrated expertise in regulatory compliance and international customs clearance for pharmaceutical shipments


