
Introduction
The stakes are high in pharmaceutical cold chain logistics. Biologics, vaccines, and specialty drugs require strict temperature control throughout transit—a single temperature excursion can compromise product efficacy and patient safety at scale. According to industry data, up to 50% of global vaccines are wasted due to cold chain failures, with specific studies showing that 25% of doses are discarded from temperature excursions alone.
As pharmaceutical supply chains grow more global and complex, selecting the right cold chain logistics provider has direct consequences for product integrity, regulatory standing, and patient outcomes. The rapid expansion of cell and gene therapies—products that often require cryogenic conditions and near-zero tolerance for deviation—has made this decision more technically demanding than ever.
This guide breaks down what pharmaceutical cold chain logistics actually requires, profiles the leading providers in 2024, and gives you a framework for evaluating them against your specific regulatory and operational constraints.
TL;DR
- Cold chain logistics maintains strict temperature ranges (2–8°C, -20°C, or -80°C) throughout storage and transport to protect pharmaceutical product integrity
- Top 2024 providers include UPS Healthcare, DHL Supply Chain, World Courier, Cryoport, and Kuehne + Nagel — each with distinct strengths by product type and region
- Selection criteria include temperature control precision, certifications (GDP, ISO, CEIV Pharma), real-time tracking technology, and global reach
- For specialty drugs and biologics, providers with cryogenic capabilities and end-to-end compliance documentation are critical
- Matching a provider to your product type, geography, and regulatory environment matters more than brand name
What Is Cold Chain Logistics in Pharmaceutical Distribution?
Pharmaceutical cold chain logistics is the unbroken, temperature-controlled supply chain required to move temperature-sensitive drugs, biologics, and vaccines from manufacturer to end user—covering storage, handling, and transport.
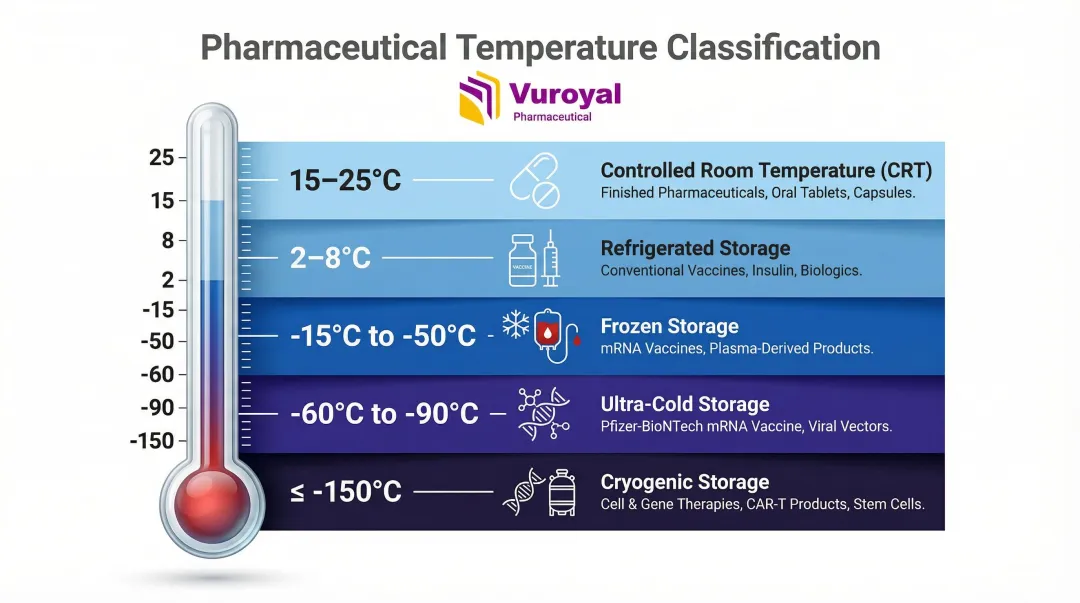
Standard Temperature Ranges
Different pharmaceutical products require specific temperature bands throughout their lifecycle:
| Temperature Range | Classification | Typical Products |
|---|---|---|
| 2–8°C | Refrigerated | Conventional vaccines, many biologics |
| -50°C to -15°C | Frozen | Moderna COVID-19 mRNA vaccine |
| -60°C to -90°C | Ultra-Cold | Pfizer-BioNTech mRNA vaccine |
| ≤ -150°C | Cryogenic | Cell and gene therapies (ATMPs), living cell preservation |
| 15–25°C | Controlled Room Temperature (CRT) | Finished pharmaceuticals; Mean Kinetic Temperature evaluated during brief excursions |
Key Regulatory Compliance Requirements
FDA, WHO, and EU Good Distribution Practice (GDP) guidelines mandate that temperature-sensitive pharmaceuticals be handled by validated cold chain processes. This means providers must maintain:
- Documented Standard Operating Procedures (SOPs)
- Continuous temperature monitoring records
- Deviation management protocols
- Calibrated monitoring equipment
- Temperature mapping of storage areas
The EU GDP Guidelines (2013/C 343/01) require wholesale distributors to maintain a quality system keeping temperature conditions within acceptable limits during transport. On the U.S. side, FDA 21 CFR Part 211 mandates written procedures for drug product storage under appropriate temperature, humidity, and light conditions—making regulatory alignment across both frameworks a baseline expectation for any compliant cold chain provider.
Top Cold Chain Logistics Providers for Pharmaceuticals in 2024
These providers were selected based on demonstrated pharmaceutical expertise, regulatory compliance, geographic reach, technology capabilities, and track record with temperature-sensitive drug shipments—not size alone.
UPS Healthcare
UPS Healthcare operates one of the world's largest healthcare logistics divisions, offering temperature-controlled air and ground freight, cGMP-compliant warehousing, and purpose-built pharmaceutical distribution networks across more than 220 countries and territories.
What Sets UPS Healthcare Apart:
The division operates more than 252 global healthcare facilities, encompassing over 19.2 million square feet of cGMP and GDP-compliant distribution space.
UPS Healthcare uses UPS Premier for advanced sensor-based tracking across three tiers: Silver (RFID for point-in-time visibility), Gold (Mesh network for near real-time visibility within 2 meters), and Platinum (Cellular sensors for real-time environmental monitoring). All shipments are monitored 24/7 by the UPS Healthcare Control Tower.
The company recently expanded its network with purpose-built cross-docking facilities in Milan, Frankfurt, and Mexico City to accelerate delivery times and bypass prolonged warehouse storage, reducing ambient exposure during transit.
| Capability | Details |
|---|---|
| Temperature Capabilities | Controlled room temperature (15–25°C), refrigerated (2–8°C), frozen (-20°C), ultra-cold (-80°C) |
| Key Certifications | IATA CEIV Pharma at 18+ global locations (Miami, New York, Shanghai, Amsterdam, Singapore); ISO 9001; ISO 13485 |
| Best For | Large-scale commercial pharmaceutical distribution, clinical-to-commercial launches, global reach across established markets |
DHL Supply Chain (Life Sciences & Healthcare)
DHL's Life Sciences & Healthcare division offers temperature-controlled warehousing, packaging, and freight forwarding with one of the broadest global footprints in pharmaceutical logistics.
Why It Stands Out:
DHL was among the early adopters of the IATA CEIV Pharma certification program and has invested in a global network of pharma-grade "Quality Centers" designed specifically for temperature-sensitive drug handling. The company delivered more than 2 billion COVID-19 vaccine doses to over 175 countries while maintaining strict regulatory compliance.
DHL operates 111 Air GxP stations worldwide, ensuring sensitive shipments are handled by Certified Life Sciences Specialists. For visibility, DHL uses its validated cloud-based platform, LifeTrack, paired with NFC SmartSensor technology, enabling 24/7 proactive monitoring, intervention management, and data analytics.
| Capability | Details |
|---|---|
| Temperature Capabilities | 15–25°C, 2–8°C, -20°C, -70°C (both active containers like Envirotainers and passive packaging solutions) |
| Key Certifications | IATA CEIV Pharma at 33+ strategic stations globally; GDP compliance across 20+ European countries |
| Best For | Multinational pharmaceutical companies needing integrated cold chain across multiple geographies, including emerging markets |

World Courier (A Cencora Company)
World Courier specializes in high-value, time-critical pharmaceutical shipments—particularly clinical trial material, investigational drugs, and specialty biologics—operating in over 50 countries with a focus on regulatory-compliant cross-border transport.
What Sets World Courier Apart:
As part of Cencora (formerly AmerisourceBergen), World Courier combines a global specialty logistics network with deep clinical trial supply expertise. The company operates over 140 global offices and 22 GxP-compliant pharmaceutical depots.
In 2023, World Courier deployed a Real-Time Location Monitoring (RTLM) solution across all multi-use packaging assets, allowing operations teams to proactively anticipate risks and intervene globally.
World Courier is a market leader in comparator drug sourcing, drawing on over 50,000 products across 30 countries. They're rapidly expanding their cryogenic network to support cell and gene therapies, operating a global network of liquid nitrogen charging centers and stations utilizing dry vapor shippers to maintain stable conditions below -150°C.
| Capability | Details |
|---|---|
| Temperature Capabilities | CRT, 2–8°C, ≤ -150°C (cryogenic liquid nitrogen vapor-phase for ATMPs) |
| Key Certifications | Station-level IATA CEIV Pharma certifications; GDP compliance across global depot network |
| Best For | Clinical trial sponsors, research organizations, and specialty drug manufacturers needing high-compliance, cross-border cold chain transport |
Cryoport
Cryoport is a purpose-built cryogenic logistics company serving the life sciences sector, with core expertise in ultra-cold and controlled-rate freezing logistics for cell and gene therapies, biologics, and vaccines—including clinical and commercial programs.
What Sets Cryoport Apart:
As of late 2025, Cryoport supported a record 760 global clinical trials and 20 commercially approved cell and gene therapies. The company was the first global supply chain company to achieve ISO 21973:2020 certification—the international standard for the transportation of cells for therapeutic use. To ensure compliance and eliminate cross-contamination, Cryoport maintains a fully segregated "Advanced Therapy Shipper" fleet dedicated exclusively to human cell and gene therapies.
Cryoport's SmartPak II condition monitoring system tracks critical shipment parameters in near real-time: location, temperature (internal/external), pressure, humidity, light exposure, shock, and orientation. Tilt monitoring is especially critical for cryogenic LN2 shippers—improper orientation can drastically reduce hold times.
All data feeds into the 21 CFR Part 11-compliant Cryoportal 2.0 platform, enabling 24/7 intervention.
| Capability | Details |
|---|---|
| Temperature Capabilities | CRT, 2–8°C, -60 to -80°C, ≤ -150°C (liquid nitrogen vapor-phase cryogenic) |
| Key Certifications | ISO 21973:2020 (first to achieve); ISO 9001:2015; ISO 13485; station-level CEIV Pharma certifications |
| Best For | Biotech companies, vaccine developers, and clinical trial sponsors shipping cell therapies, biologics, or cryogenic materials |

Kuehne + Nagel (HealthChain)
Kuehne + Nagel's HealthChain (formerly KN PharmaChain) offers integrated cold chain logistics solutions including air, sea, and ground freight with pharma-specific temperature-control packaging, real-time monitoring, and dedicated pharmaceutical lanes globally.
Notable Capabilities:
In 2023, Kuehne + Nagel became the first logistics company to achieve over 100 IATA CEIV Pharma-certified stations, covering 66 countries across all seven continents. The company now operates HealthChain across more than 270 certified locations worldwide.
Kuehne + Nagel uses a dedicated HyperCare team that monitors shipments 24/7/365. Acting as an "immune system" for the supply chain, this team applies predictive analytics and lane risk assessments to react at the first sign of a temperature deviation.
The company has also established dedicated temperature-controlled corridors (+2 to +8°C and +15 to +25°C) that shift from active to sustainable passive packaging where appropriate—reducing both carbon emissions and operational costs.
| Capability | Details |
|---|---|
| Temperature Capabilities | CRT (15–25°C), 2–8°C, -20°C, -70°C (multi-modal with both active and passive packaging) |
| Key Certifications | 100+ IATA CEIV Pharma stations; WHO GDP; ISO 9001; ISO 14001; ISO 45001 |
| Best For | Pharmaceutical manufacturers and distributors needing lane-qualified, multimodal cold chain with strong data documentation for regulatory filings |
How We Chose the Best Cold Chain Logistics Providers
Providers were assessed across six criteria:
- Temperature control precision and validated performance data
- Geographic coverage and network depth
- Regulatory certifications (GDP, ISO, CEIV Pharma)
- Real-time monitoring technology
- Clinical and commercial track record
- Capacity to handle specialty shipments such as biologics and cryogenics
Common Mistakes Buyers Make
Organizations often over-rely on brand size rather than evaluating pharmaceutical-specific certifications. MHRA data indicates that temperature excursions are a significant cause of non-compliance, affecting approximately 30% of inspections. Common failures include:
- Missing or outdated calibration records for monitoring devices
- Failure to properly investigate and document temperature alarms (CAPA failures)
- Inadequate documentation, which contributes to nearly 40% of GDP non-compliance findings

Buyers frequently fail to validate that a provider's temperature-control documentation meets the exact regulatory standards (FDA, EMA, ICH Q10) required for their product and target market. They also neglect to assess deviation management and excursion response protocols before signing contracts.
Beyond Standard Freight Forwarding
For organizations sourcing specialty pharmaceutical substances internationally—such as vaccine adjuvants, reference listed drugs, or investigational compounds—end-to-end cold chain compliance and customs expertise are equally critical.
VuRoyal Pharmaceutical addresses this gap directly. As an FDA-registered pharmaceutical wholesaler, VuRoyal pairs cold chain transportation with regulatory guidance for cross-border procurement. The company holds drug business licenses, quality management certifications, and customs declaration credentials—covering imported drug approval registration, import filing, international procurement, customs clearance, and transportation under one contract.
VuRoyal's GDP Standard Operation Protocol is built on USP guidelines, Title 21 CFR, PDMA, and DSCSA requirements. Third-party compliance solutions reinforce federal and state regulatory adherence. Documentation provided includes:
- Imported Drug Approval Documents
- Legal source verification
- Complete chain-of-custody records
Conclusion
Choosing a cold chain logistics provider for pharmaceuticals requires more than comparing rates or brand recognition. The right partner must demonstrate validated temperature-control performance, regulatory compliance, and the resources and experience to handle your specific product type and target markets.
When evaluating candidates, prioritize providers that offer:
- Full chain-of-custody documentation at every handoff point
- Proactive deviation management with documented corrective actions
- Scalable capacity for clinical-to-commercial transitions and market expansions
Under ICH Q10 guidelines, the pharmaceutical company retains ultimate responsibility for outsourced quality activities — making rigorous provider assessment and ongoing monitoring non-negotiable.
That accountability extends to every link in the chain, including your distribution partners. For research organizations and pharmaceutical companies sourcing specialty drugs, biologics, or vaccine adjuvants across borders, VuRoyal Pharmaceutical offers specialty drug logistics with cold chain transportation and regulatory support for international procurement and cross-border distribution. Contact VuRoyal to discuss your supply chain requirements.
Frequently Asked Questions
What is cold chain logistics in pharmaceuticals?
Pharmaceutical cold chain logistics refers to the temperature-controlled storage and transport system that maintains drug products within required temperature ranges (e.g., 2–8°C, -20°C, ≤ -150°C) from manufacturer to end user to preserve efficacy and safety.
What certifications should a pharmaceutical cold chain provider have?
Look for GDP compliance, IATA CEIV Pharma certification for air freight, ISO 9001 quality management, and FDA 21 CFR Part 211 compliance for U.S.-regulated shipments. For advanced therapies such as biologics and cell/gene products, ISO 21973 certification is also required.
What is the difference between active and passive cold chain packaging?
Active systems use powered refrigeration (e.g., electric containers) to maintain temperature, while passive systems use insulated packaging with phase-change materials (dry ice, gel packs). Active systems offer longer duration and more precise control but at higher cost.
What temperature range is required for storing and shipping vaccines?
Most conventional vaccines require 2–8°C (refrigerated), while mRNA vaccines require -20°C to -80°C (frozen or ultra-cold). The Moderna COVID-19 vaccine requires -50°C to -15°C, while Pfizer-BioNTech requires -90°C to -60°C.
How do you verify a cold chain provider's compliance for pharmaceutical shipments?
Request GDP documentation, lane qualification studies, temperature excursion SOPs, and references from pharma clients. Confirm third-party or regulatory audits of the provider's facilities, and review their CAPA implementation records for how they resolve deviations.
Why is cold chain integrity especially important for biologics and vaccine adjuvants?
Biologics and adjuvants like QS-21 are highly sensitive to temperature deviations that cause protein degradation, loss of immunogenicity, or aggregation. Free QS-21, for instance, exceeds acceptable hydrolysis limits after just one month at 45°C — a deviation that directly compromises both patient safety and regulatory standing.


