
Introduction
Pharmaceutical supply chains in 2025 face mounting pressure from every direction. Geopolitical tensions, post-pandemic vulnerabilities, and stricter global regulations are converging with the rapid rise of biologics and specialty drugs that demand precision logistics. The industry loses approximately $35 billion annually due to temperature-controlled logistics failures alone, while over 270 active drug shortages persist across critical therapeutic categories.
Pharma companies need supply chains that are faster, more transparent, and more resilient — and the gap between laggards and leaders is widening fast. This guide covers the AI tools, automation platforms, and distribution strategies helping research organizations, clinical trial sponsors, and manufacturers close that gap in 2025.
TLDR
- Pharma SCM in 2025 is a strategic business priority, not just logistics—driven by AI, automation, and resilience planning
- Key trends shaping 2025: AI-driven demand forecasting, blockchain traceability, digital twins, cold chain innovation, and sustainability compliance
- Leading solutions: SAP IBP, Oracle SCM Cloud, TraceLink, Veeva Vault Quality, and Blue Yonder Luminate
- When evaluating platforms, prioritize regulatory compliance, cold chain support, system interoperability, and global scalability
- Specialty drugs and biologics require cold chain-certified distribution partners—software alone won't close the compliance gap
Why Pharma Supply Chain Management Matters More Than Ever in 2025
Pharmaceutical supply chain management spans procurement, manufacturing, quality control, cold chain logistics, regulatory compliance, and last-mile distribution. In 2025, the convergence of persistent drug shortages, cold chain failures, and tightening global regulations has elevated it from a back-office concern to a boardroom priority.
The global pharma SCM market is valued at $3.4 billion in 2025 and projected to reach $7.3 billion by 2034, reflecting an 8.8% CAGR. That growth is fueled by mounting pressure: the FDA reported 271 active drug shortages in early 2024, each affecting at least half a million consumers. The financial toll is steep — drugs in shortage see an average 16.6% price increase while sales volume drops 28% to 35%.
Beyond shortages, temperature-controlled logistics failures cost the industry $35 billion annually, with up to 50% of global vaccines wasted due to cold chain breakdowns. These disruptions don't just impact margins — they delay clinical trials, force treatment substitutions, and directly compromise patient safety.
Today's pharma SCM must balance three competing pressures simultaneously:
- Cost-efficiency: Reducing waste, optimizing inventory, and minimizing logistics expenses
- Regulatory compliance: Meeting FDA, EMA, DSCSA, and GMP requirements across multiple jurisdictions
- Patient access: Ensuring uninterrupted availability of life-critical medications
Companies that master this balance are better positioned to absorb disruptions, scale into new markets, and protect both patients and margins — which is precisely why AI and automation are reshaping how pharma supply chains are built and managed today.
2025 Trends Reshaping Pharma Supply Chains: AI, Automation & Beyond
AI and Machine Learning for Demand Forecasting
Traditional demand planning built on spreadsheets and historical averages can no longer keep pace with today's supply volatility. AI-powered forecasting uses machine learning algorithms like Random Forest and XGBoost to predict demand fluctuations, flag supply disruptions before they occur, and automate replenishment decisions.
Machine learning models outperform traditional ARIMA methods by 12% to 15% in forecast accuracy while reducing planner workloads by 20% to 30%. By 2030, 70% of large organizations will adopt AI-based forecasting, according to Gartner.
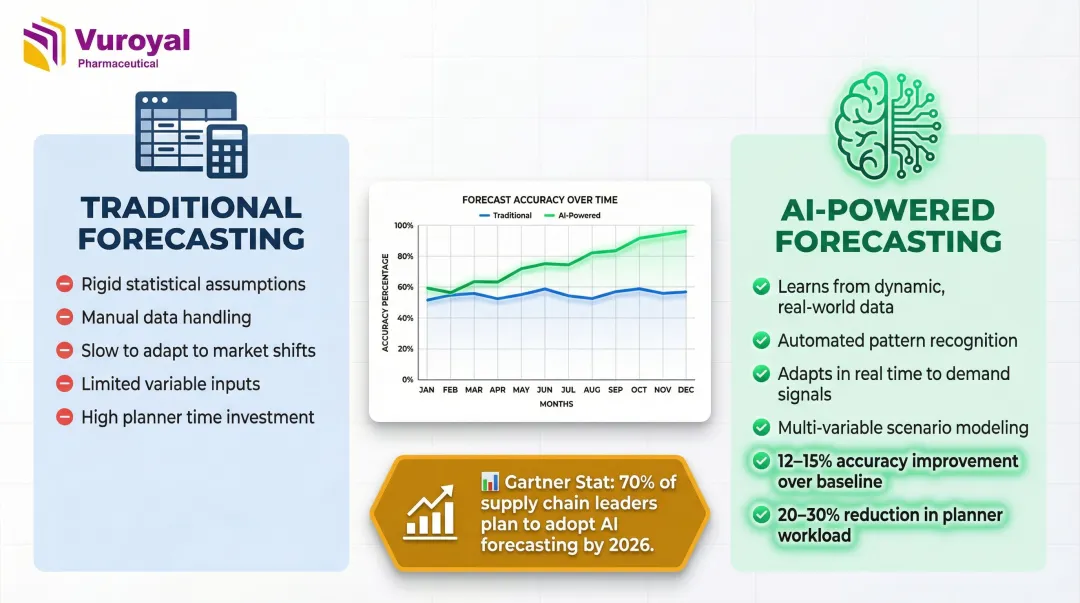
AI doesn't just improve accuracy—it enables adaptive, real-time decision-making. Systems analyze diverse data sources including prescription trends, seasonal patterns, regulatory changes, and external market signals to dynamically adjust forecasts. This reduces both costly stockouts and excess inventory holding costs.
Digital Twins and Simulation
Digital twins create real-time virtual replicas of physical supply chains, allowing companies to simulate new distribution routes, stress-test operations during disruptions, and optimize performance without halting real-world operations.
The digital twins market for pharmaceutical manufacturing is valued at $1.3 billion in 2025 and forecast to reach $8.5 billion by 2032 at a 30.2% CAGR. By 2028, an estimated 63% of pharma production lines will use digital twins — up from just 17% in 2025.
Real-world results validate the technology:
- Pfizer achieved 40% faster operator onboarding using VR environments replicating shop floors during vaccine scale-up
- GSK reduced physical "wet" experiments by creating real-time simulations of entire vaccine manufacturing processes
- AstraZeneca scaled digital apps across 120 packaging lines in 17 global sites, achieving a 29% reduction in changeover time
Digital twins enable scenario planning that would be impossible with physical systems—testing new suppliers, modeling pandemic surges, or evaluating nearshoring strategies without disrupting operations.
Blockchain for Traceability and Anti-Counterfeiting
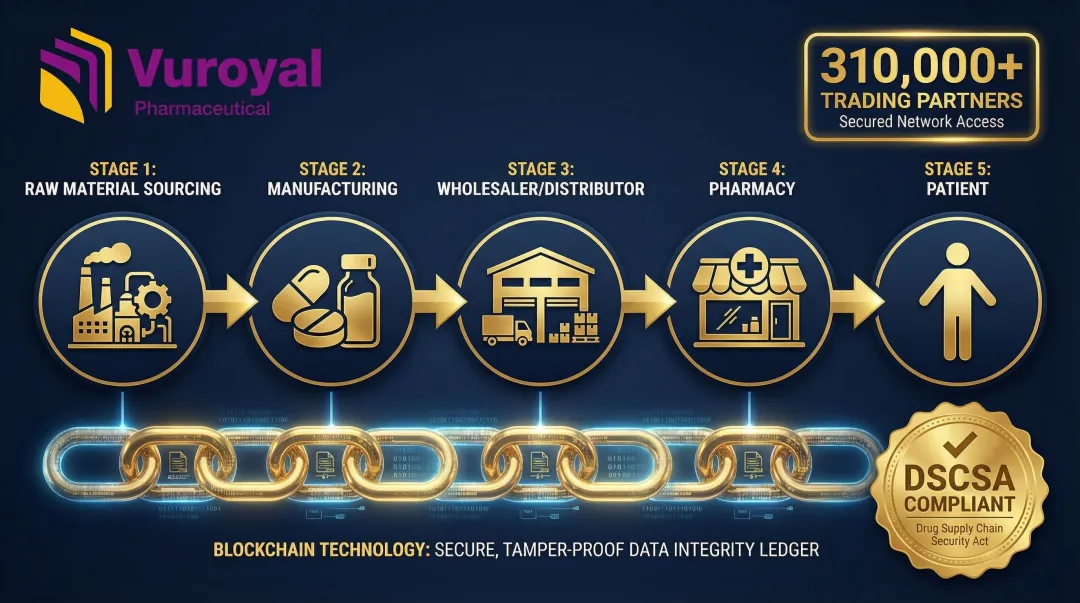
Counterfeit medicines cost countries $30.5 billion annually, with at least 1 in 10 medicines in low- and middle-income countries being substandard or falsified. Blockchain creates immutable, end-to-end records from raw material sourcing to final delivery, verifying drug authenticity at every step.
The U.S. Drug Supply Chain Security Act (DSCSA) mandates interoperable, electronic tracing of prescription drugs at the package level. Following a 1-year stabilization period that ended November 27, 2024, full enforcement began rolling out in 2025 — making compliant traceability infrastructure a practical necessity, not just best practice.
Legacy point-to-point EDI integrations cost $5,000 to $15,000 per partner relationship and take 4-8 weeks to implement—making them unscalable for DSCSA. Network-based solutions solve this:
- MediLedger completed an FDA pilot with 24 industry leaders (including Pfizer, Novartis, Sanofi) proving blockchain feasibility
- TraceLink connects 310,000+ trading partners and processes over 7 billion regulated transactions annually through its "Integrate Once, Interoperate with Everyone" network model
Blockchain doesn't just meet compliance requirements—it builds trust across global supply networks while substantially reducing integration costs.
Cold Chain Innovation and IoT Monitoring
With 80% of pharmaceutical products now requiring temperature control, cold chain failures remain one of the costliest challenges in pharma logistics. The industry loses $35 billion annually to temperature-controlled logistics failures, with up to 50% of global vaccines wasted due to cold chain breakdowns.
Real-time IoT sensors now track temperature and humidity throughout transit, allowing logistics teams to intervene before excursions destroy high-value biologics. The global cold chain monitoring market reflects this urgency: growing from $8.31 billion in 2025 to $15.04 billion by 2030 at a 12.6% CAGR.
New technologies address both financial losses and environmental impact:
- Predictive analytics identify potential failures before they occur
- Cloud platforms provide real-time visibility across entire cold chains
- Eco-friendly packaging maintains temperature integrity while cutting environmental footprint
- Automated alerts enable rapid response to temperature deviations, with configurable thresholds by product type
For biologics, vaccines, and specialty drugs—where a single temperature excursion can render millions of dollars in product unusable—these innovations are no longer optional.
Supply Chain Resilience and Nearshoring
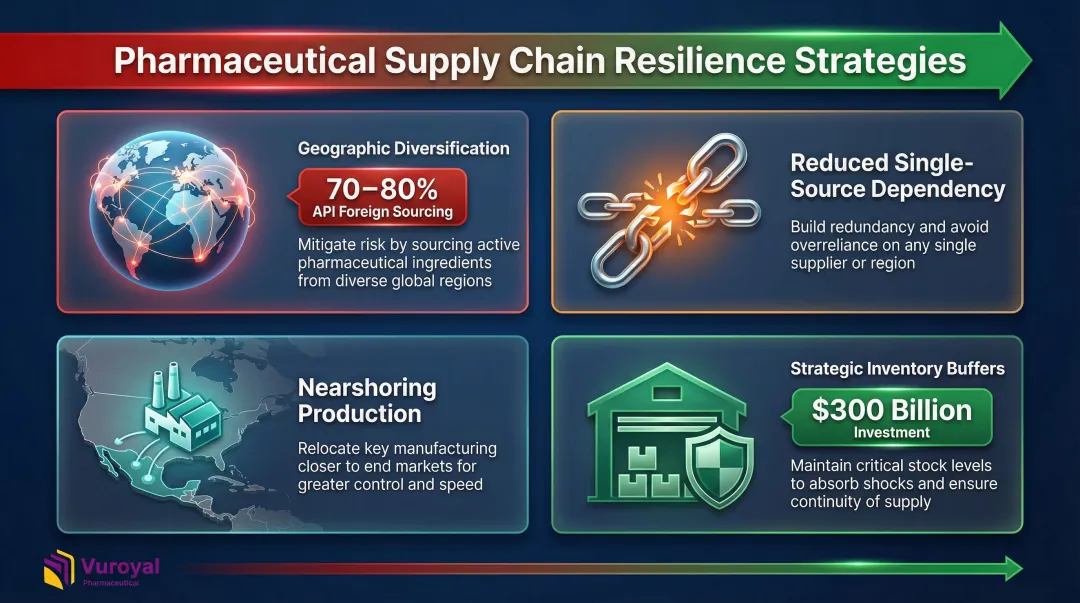
The U.S. relies on foreign sources for 70% to 80% of its Active Pharmaceutical Ingredients (APIs), with China controlling 41% of key starting materials and holding near-monopolies (over 99%) on critical inputs like sulfonamide-class antibiotics.
Two regulatory pressures are accelerating change. The BIOSECURE Act (H.R.8333) prohibits federally funded entities from using biotechnology linked to foreign adversaries, while threats of 100% tariffs on imported branded drugs have pushed manufacturers to act faster than planned.
The response is already underway: over $300 billion in investments have been announced by major drugmakers including Eli Lilly, J&J, and AstraZeneca to build new domestic and nearshore production plants. Companies are:
- Diversifying manufacturing across multiple geographies
- Reducing single-source dependency for critical APIs
- Nearshoring production to reduce geopolitical risk
- Building strategic inventory buffers for essential medicines
For supply chain leaders, the takeaway is direct: single-geography sourcing for critical materials carries risks that financial hedging alone cannot offset.
Top 5 Pharma Supply Chain Management Solutions for 2025
These five solutions were selected based on regulatory capabilities, AI-driven functionality, pharma-sector adoption, scalability, and ability to address the specific complexity of pharmaceutical and life sciences supply chains.
SAP Integrated Business Planning (IBP)
SAP IBP is a cloud-based, enterprise-grade platform that unifies sales, demand planning, inventory management, and supply chain execution. Widely adopted by large pharmaceutical manufacturers, it delivers real-time analytics and AI-powered forecasting capabilities.
What sets it apart from general SCM platforms:
- Deep integration with SAP's broader ERP ecosystem
- Scenario-based planning tools for managing multi-market pharmaceutical portfolios
- Capacity planning for complex batch production environments
Moderna leveraged SAP IBP during COVID-19 vaccine scale-up to manage unprecedented demand volatility.
| Attribute | Details |
|---|---|
| Key Strength | AI-driven demand forecasting and real-time supply chain visibility across global operations |
| Best Suited For | Large pharmaceutical manufacturers with complex, multi-site production and distribution networks |
| Regulatory Fit | Integrates with GxP-compliant workflows and FDA/EMA reporting requirements |
Oracle Supply Chain Management (SCM) Cloud
Oracle SCM Cloud is a comprehensive suite covering procurement, product lifecycle management, logistics, order management, and supplier collaboration. Used across pharma and biotech companies globally, it's built to grow with the business as operations expand.
Three capabilities distinguish it from competing platforms:
- Native IoT and AI integration for predictive decision-making
- Cloud-native architecture that scales with production volume
- Supply chain visibility tools critical for pharmaceutical traceability and compliance reporting
Companies like 10x Genomics and Guardant Health rely on Oracle SCM Cloud for their operations.
| Attribute | Details |
|---|---|
| Key Strength | End-to-end supply chain visibility with IoT integration and AI-powered operational insights |
| Best Suited For | Mid-to-large pharma and biotech companies seeking a scalable, cloud-first SCM platform |
| Regulatory Fit | Supports compliance tracking, quality management, and global regulatory reporting frameworks |
TraceLink
TraceLink is a pharma-specific supply chain platform built around serialization, traceability, and supply network digitization. Purpose-built to help pharmaceutical companies meet DSCSA requirements in the U.S. and comparable serialization mandates globally.
The platform distinguishes itself through its network-based architecture — connecting manufacturers, distributors, 3PLs, and pharmacies on a single shared platform rather than point-to-point integrations. Compliance automation is built in, not bolted on. TraceLink connects 310,000+ trading partners and processes over 7 billion regulated transactions annually.
| Attribute | Details |
|---|---|
| Key Strength | Purpose-built pharmaceutical serialization, traceability, and DSCSA compliance automation |
| Best Suited For | Pharmaceutical manufacturers, wholesalers, and distributors needing regulatory-grade supply chain traceability |
| Regulatory Fit | Designed specifically for FDA DSCSA, EU FMD, and other global drug traceability regulations |
Veeva Vault Quality Suite
Veeva Vault Quality is a life sciences–specific cloud platform that integrates quality management, document control, and supplier quality oversight directly into supply chain workflows. Widely adopted by biopharma, vaccine developers, and specialty drug manufacturers.
Unlike general enterprise software adapted for pharma, Veeva Vault Quality was built exclusively for life sciences from the ground up. Key capabilities include:
- Direct linkage between quality events and live supply chain operations
- GMP compliance documentation support for regulated manufacturing
- Supplier quality oversight integrated natively into procurement workflows
Companies like Roche and Celularity use Veeva Vault Quality.
| Attribute | Details |
|---|---|
| Key Strength | Life sciences–native quality management integrated directly with supply chain and supplier oversight |
| Best Suited For | Biopharma companies, vaccine developers, and specialty drug manufacturers with complex quality compliance needs |
| Regulatory Fit | Purpose-built for GMP, GxP, and FDA/EMA quality system requirements |
Blue Yonder Luminate Supply Chain
Blue Yonder Luminate is an AI-first supply chain platform offering end-to-end demand planning, logistics orchestration, and warehouse management. Increasingly adopted in the pharmaceutical sector for its autonomous planning capabilities and predictive analytics engine.
Three capabilities make it particularly relevant for pharmaceutical supply chains:
- AI/ML demand forecasting models that adapt in real time to market signals
- Cold chain logistics orchestration for temperature-sensitive pharmaceutical products
- Microservices-based architecture that integrates with existing ERP and manufacturing systems
Blue Yonder achieves up to 12% improvement in forecast accuracy using advanced machine learning.
| Attribute | Details |
|---|---|
| Key Strength | Autonomous AI-driven supply chain planning with real-time demand sensing and logistics orchestration |
| Best Suited For | Pharmaceutical companies prioritizing AI-powered demand forecasting and adaptive supply chain management |
| Regulatory Fit | Supports audit trails and compliance-ready data management for regulated pharmaceutical environments |
How We Chose These Pharma SCM Solutions
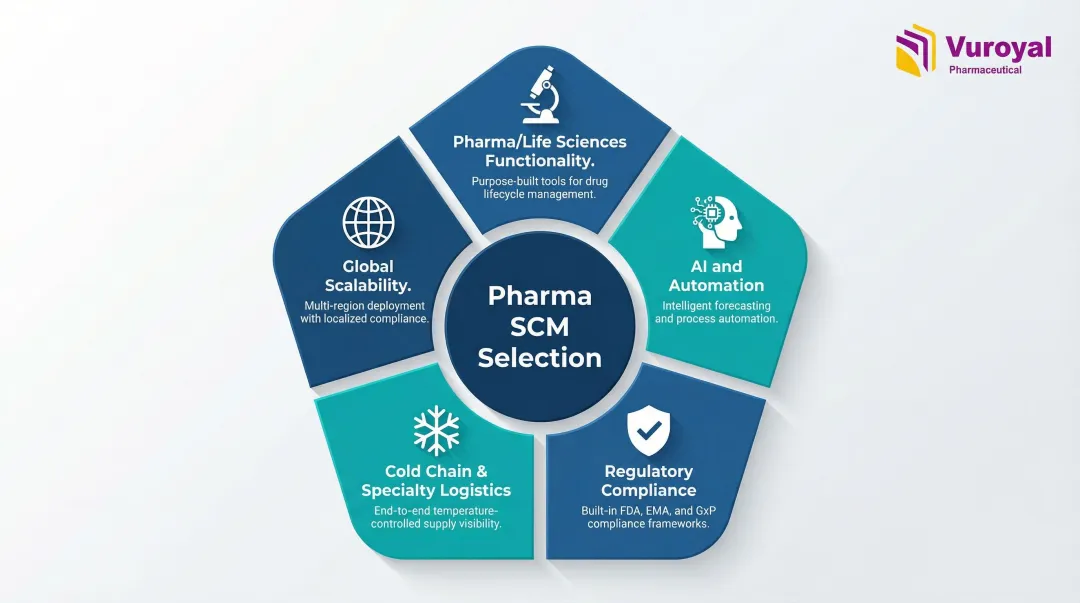
Solutions were assessed on five criteria:
- Pharma/life sciences functionality — depth of domain-specific features vs. generic ERP capabilities
- AI and automation capabilities — demand forecasting, serialization, and workflow automation
- Regulatory compliance support — alignment with FDA, DSCSA, and GMP requirements
- Cold chain and specialty logistics — temperature-controlled handling and serialized tracking
- Global scalability — ability to support multi-market operations and cross-border procurement
A common mistake: Buyers often prioritize general-purpose enterprise software with extensive features over specialized tools that fit the unique regulatory and quality demands of pharmaceutical supply chains. A pharmaceutical-grade solution or compliant distribution partner often outperforms a generic platform in practice.
That distinction matters most for organizations managing specialty drugs, biologics, or complex procurement — including clinical trial sponsors and vaccine developers. For these use cases, software selection is only part of the equation.
VuRoyal Pharmaceutical is an FDA-registered wholesaler that provides end-to-end supply chain services for specialty pharmaceutical procurement: cold chain logistics, customs clearance, and free regulatory guidance for cross-border pharmaceutical procurement.
Conclusion
The pharma supply chain landscape in 2025 demands a dual strategy: the right technology platform to drive visibility, forecasting, and compliance, paired with the right operational partner to ensure reliable, compliant delivery of pharmaceutical substances.
When evaluating SCM solutions and partners, prioritize substance over marketing:
- Proven regulatory track record across FDA, DEA, and applicable international frameworks
- Scalability for your product categories — biologics, specialty drugs, and vaccine adjuvants each carry distinct cold chain and handling requirements
- Domestic and global supply capability, not just regional reach
These criteria matter more than feature lists or brand recognition.
For organizations whose needs include specialty pharmaceutical procurement, cold chain distribution, or international importation, those criteria point toward a narrow field of qualified partners. VuRoyal Pharmaceutical — an FDA-registered, Massachusetts-approved wholesaler — is built precisely around that intersection, offering certified end-to-end supply chain services and free regulatory guidance for imported drug registration and filing.
Frequently Asked Questions
Frequently Asked Questions
What is pharmaceutical supply chain management?
Pharma SCM covers the full journey from sourcing raw materials through manufacturing, quality assurance, and distribution to patients. Unlike general supply chains, it operates under strict regulatory oversight because product failures directly affect patient safety.
How is AI being used in pharma supply chain management in 2025?
AI powers demand forecasting, predictive disruption alerts, automated replenishment, and real-time supply chain visibility. It moves planning beyond historical averages, enabling real-time decisions that improve forecast accuracy by 12% to 15%.
What are the biggest challenges in pharmaceutical supply chain management today?
The most pressing challenges today include:
- Geopolitical disruptions cutting off key raw material sources
- Serialization and traceability compliance under frameworks like DSCSA
- Cold chain integrity for biologics and specialty drugs
- Counterfeit products infiltrating global supply networks
What is cold chain management in pharma and why does it matter?
Cold chain management refers to temperature-controlled logistics required for biologics, vaccines, and other temperature-sensitive pharmaceuticals. Failures in cold chain integrity can render products ineffective or unsafe — and the industry loses an estimated $35 billion annually to temperature excursions, which is why real-time IoT monitoring has become standard practice.
How do serialization requirements like DSCSA affect pharma supply chains?
The U.S. Drug Supply Chain Security Act (DSCSA) requires pharmaceutical manufacturers, wholesalers, and dispensers to serialize, track, and trace drug products at the unit level. This adds compliance complexity but it enables reliable drug authenticity verification at every handoff — a direct improvement to patient safety outcomes.
What should organizations look for when selecting a pharma supply chain management solution?
Key selection criteria include:
- Pharma-specific compliance support (GMP, DSCSA, FDA alignment)
- AI-driven demand planning and disruption forecasting
- Cold chain logistics and IoT monitoring capabilities
- Interoperability with existing ERP systems
- Scalability for international procurement and multi-market distribution


