QS-21 Vaccine Adjuvant
- 95%+ efficacy in Shingrix shingles vaccine
- 200–400M human doses annual capacity
- FSC-certified sustainable bark sourcing



GMP Certified
EXCiPACT Certified
FDA-Registered
QS-21 Products encompass a range of GMP-certified vaccine adjuvants and pharmaceutical distribution services centered on QS-21, a potent saponin derived from Quillaja saponaria Molina. Used in 5 globally approved vaccines — including Shingrix, Arexvy, and Mosquirix — QS-21 enhances both antibody production and cellular immunity, supporting human vaccine development, clinical trials, and veterinary applications worldwide.
Talk to our experts for custom solutions and tailored guidance.
Apply Filters




QS-21 is a highly purified saponin adjuvant derived from the bark of Quillaja saponaria Molina, the Chilean soapbark tree. With a molecular weight of 1990 Da, it is classified as an acylated triterpene glycoside and is recognized as one of the most potent immunological adjuvants globally, used in five approved vaccines including Shingrix, Arexvy, Mosquirix, and COVID-19 vaccines.
Talk to our experts for custom solutions and tailored guidance.
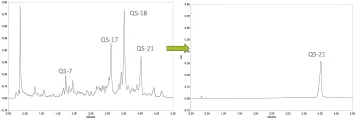
QS-21 adjuvants power approved vaccines for shingles, RSV, malaria, and COVID-19, with 20+ candidates in clinical trials.

Supports Phase I–III trials for HIV, cancer, Alzheimer's, herpes, and pneumococcal vaccine candidates requiring GMP-grade adjuvants.

End-to-end compliant distribution of FDA reference drugs and vaccine adjuvants to hospitals, clinics, and research organizations globally.

Veterinary-grade adjuvants including QA-21, Vet-sap, and Ultra support animal health vaccine innovation and efficacy enhancement.
Talk to our experts for custom solutions and tailored guidance.
As the sole authorized partner of Desert King International in China since 2023, VuRoyal provides unmatched access to the world's leading GMP-grade QS-21 adjuvant.
From FDA registration and IPEC GMP certification to import filing and customs clearance, VuRoyal ensures fully compliant, seamless global pharmaceutical supply chain management.
Backed by decades of pharmaceutical R&D experience, VuRoyal offers free regulatory guidance, expert consultation, and scientific forums to accelerate vaccine development outcomes.
Submit your inquiry and our team will respond with pricing, regulatory guidance, and tailored supply chain solutions within one business day.
For immediate assistance, feel free to give us a direct call at +1 617-870-2690 You can also send us a quick email at
For immediate assistance, feel free to give us a direct call at +1 617-870-2690 You can also send us a quick email at