
Introduction
The bio-pharma logistics sector faces unprecedented challenges: approximately $35 billion is lost annually due to temperature excursions and cold chain failures, while up to 50% of vaccines are discarded globally because of logistics issues. As biologics and cell and gene therapies proliferate—with 48 FDA-approved cellular therapies and over 3,200 ongoing clinical trials—the stakes have never been higher for manufacturers, distributors, and clinical sponsors.
In this space, precision, compliance, and speed are non-negotiable. The forces reshaping bio-pharma logistics—cold chain innovation, AI-driven supply chain tools, regulatory serialization mandates, and geopolitical dual-sourcing pressures—demand that stakeholders make smarter, more resilient decisions. Understanding these trends is what separates organizations that protect product integrity from those that don't.
TL;DR
- The global biopharma logistics market will reach $234.58 billion by 2031, driven by biologics expansion and rising regulatory complexity
- Cold chain failures cost the industry $35 billion annually — and 50% of new medicines now require temperature control
- AI and IoT are shifting supply chains from reactive monitoring to real-time intervention before failures occur
- Geopolitical pressures accelerate dual-sourcing, with 61% of executives planning nearshoring or domestic manufacturing
- DSCSA enforcement now centers on exception management, while blockchain moves from pilot programs into live operations
Key Trends Reshaping Bio-Pharma Logistics
The biopharma logistics landscape is shifting across five dimensions simultaneously — patient needs, regulatory pressure, and technological capability are all moving at once. Understanding each trend individually helps clarify where supply chain investment is headed and why.
Trend 1: Advanced Cold Chain Logistics and Temperature Control Innovation
Approximately 70% of top-selling pharmaceutical products require temperature-controlled logistics, and the stakes are rising. Medicines requiring cold storage accounted for 35% of the pharmaceutical market in 2022, up from 26% in 2017. Projections indicate that roughly 50% of all new medicines launched in the next five years will require cold storage and distribution. The global biopharma cold chain logistics market was valued at $18.23 billion in 2024 and is projected to reach $51.58 billion by 2034, reflecting a 10.98% CAGR.
Yet despite massive infrastructure investments, the industry loses $35 billion annually to temperature excursions and logistics failures. Those losses span spoiled products, clinical trial delays, wasted logistics costs, and root-cause analysis expenses.
Next-generation solutions have moved from passive monitoring to active intervention:
- IoT-enabled sensors like Controlant's real-time monitoring devices have reduced temperature deviations by 25%
- Cryogenic containers such as Cryoport Express Combo Liquid Nitrogen Dry Vapor Shippers maintain temperatures below -150°C for up to 10 days, equipped with Smartpak™ Condition Monitoring Systems tracking GPS location, temperature, orientation, and shock events
- Reusable pallet shippers like Cold Chain Technologies' CCT Tower Elite offer 120+ hours of precise temperature control (-60°C to +20°C) with integrated IoT monitoring
- Active refrigerant containers such as Envirotainer RKN e1 maintain +2°C to +8°C even in extreme ambient conditions

Companies like VuRoyal provide specialty drug logistics with cold chain transportation for temperature-sensitive products, including vaccine adjuvants and reference listed drugs for clinical trials. Their model covers international procurement, customs clearance, and final delivery — keeping product integrity intact across every handoff.
Trend 2: Digital Transformation — AI, IoT, and Real-Time Supply Chain Visibility
The global AI in pharmaceutical market is projected to reach $16.49 billion by 2034, yet enterprise adoption remains fragmented. A Gartner survey reveals that only 23% of supply chain leaders report having a formal supply chain AI strategy in place. Most Chief Supply Chain Officers focus on "project-by-project" short-term wins, which produces a patchwork of disconnected tools that can't scale or share data effectively.
Despite strategic gaps, specific AI and IoT applications are gaining measurable traction:
- Demand forecasting and waste reduction: 40% of companies prioritize AI for these applications
- Automated cold chain monitoring: 69% of pharmaceutical companies have implemented AI-driven automated alerts for real-time cold chain logistics monitoring
- Predictive analytics: The global cold chain monitoring market is expected to grow from $8.31 billion in 2025 to $15.04 billion by 2030 (12.6% CAGR)
Real-world deployments show what this looks like operationally. FedEx Surround uses AI and SenseAware ID sensors that transmit precise package location data every two seconds via Bluetooth Low Energy, providing near real-time global visibility and predictive disruption analytics. UPS Premier deploys cellular and mesh sensor trackers for priority handling, real-time visibility, and proactive recovery for critical healthcare shipments.
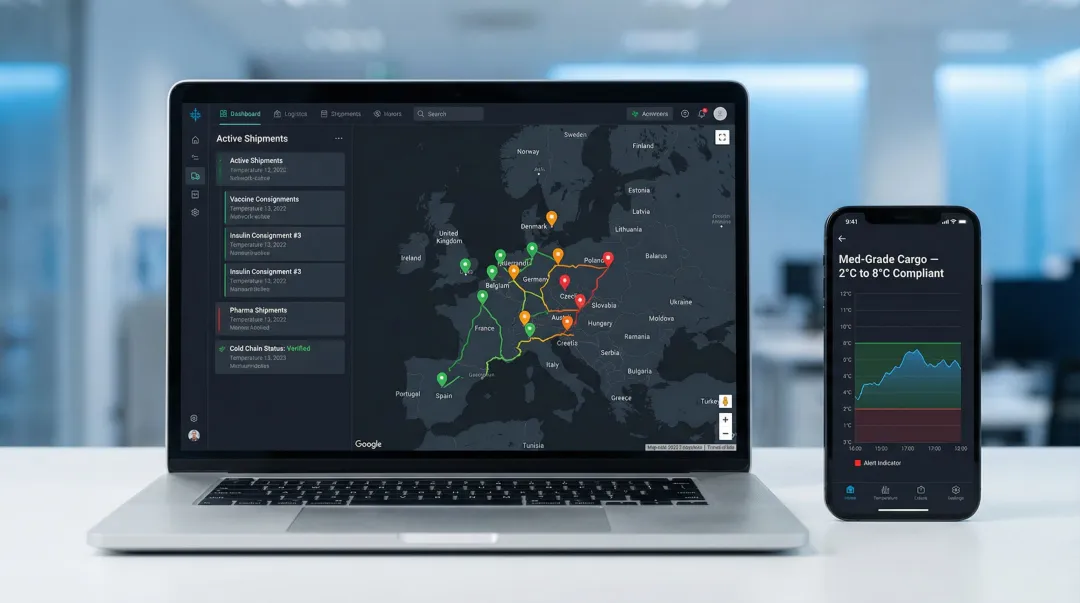
These platforms enable logistics providers to shift from reactive problem-solving to proactive intervention—predicting and preventing excursions before product degradation occurs.
Trend 3: Supply Chain Diversification and Dual-Sourcing
The global supply of Active Pharmaceutical Ingredients (APIs) remains highly concentrated. While India holds 48% of total active API Drug Master Files (DMFs), China's manufacturing capacity is surging. In 2024, China surpassed India as the top holder of new API DMF filings, capturing 45% of the total. The EU depends on Asia for 70% of its APIs and 79% of its medicine precursors.
Western governments are responding through legislation. The U.S. BIOSECURE Act, included in the FY 2026 National Defense Authorization Act (NDAA), prohibits federal agencies from contracting with biotechnology companies of concern. The Biomedical Advanced Research and Development Authority (BARDA) also released a Request for Project Proposals with approximately $200 million in funding to strengthen domestic manufacturing of essential medicines through the BioMaP Consortium.
In Europe, the Commission proposed the Critical Medicines Act in March 2025 to improve availability and production of critical medicines within the EU through strategic projects and collaborative procurement. The Health Emergency Preparedness and Response Authority (HERA) is analyzing supply chain vulnerabilities for 268 critical medicines to guide remedial actions.
The industry is responding rapidly: A 2025 index reveals that 61% of biopharma executives plan to increase domestic sourcing and/or onshore drug manufacturing over the next 12 months. Furthermore, 39% of respondents facing tariff impacts are pursuing dual sourcing strategies, and 33% are developing supplier nearshoring plans.
Leading biopharma companies are mapping supply chain vulnerabilities, qualifying backup suppliers, and investing in regional manufacturing hubs to build resilience without sacrificing regulatory compliance.
Trend 4: Specialized Logistics for Cell and Gene Therapies
Cell and gene therapies (CGTs) represent one of the fastest-growing and most logistically complex product categories. The global cell and gene therapy third-party logistics market was valued at $1.42 billion in 2024 and is expected to grow at a CAGR of 20.7% to reach $4.37 billion by 2030.
Autologous therapies require patient-specific supply chains, ultra-low temperature (ULT) storage below -80°C, and cryogenic conditions below -150°C. These temperature thresholds alone set CGTs apart from standard cold chain handling.
Beyond temperature, AABB standards mandate strict Chain of Identity (COI) and Chain of Custody (COC) tracking — the permanent, transparent association of a therapy's unique identifiers from tissue procurement through the full product lifecycle.
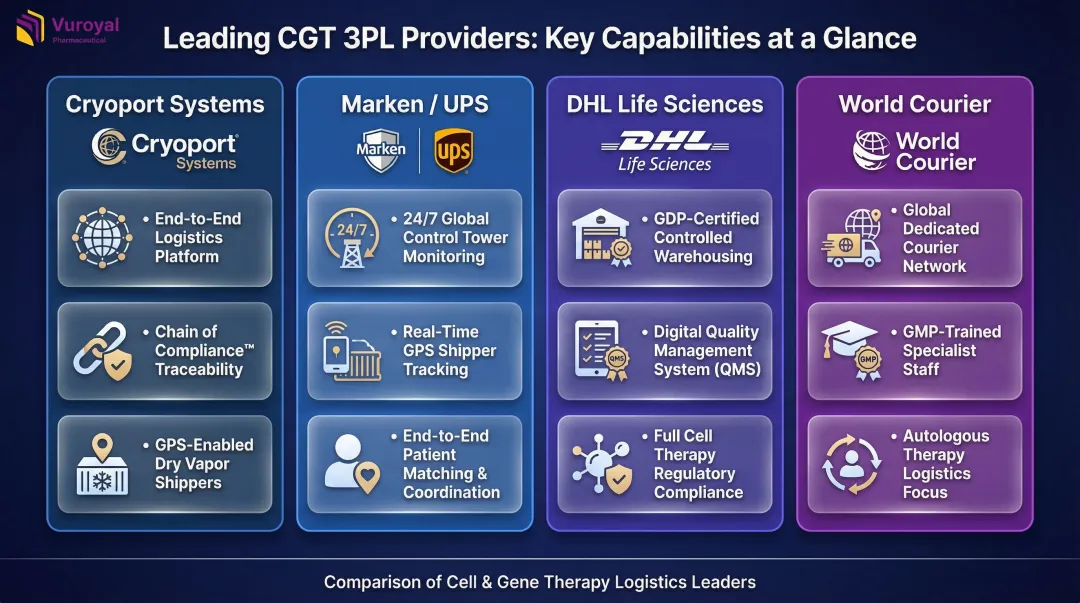
3PLs are investing heavily in dedicated infrastructure:
- Cryoport Systems: Operates the Cryoportal® logistics management platform with Chain of Compliance® traceability and GPS-tracked dry vapor shippers
- Marken (UPS): Provides 24/7 Cell & Gene Control Tower, GPS-enabled shippers, and patient matching workflows
- DHL Life Sciences: Maintains GDP-certified warehouses with digital quality management systems (QMS) for cell therapy compliance
- World Courier: Operates a global network of dedicated couriers with GMP training emphasis for autologous therapies
Orchestration platforms like TrakCel's OCELLOS (operational at 250+ sites globally) and Vineti's Personalized Therapy Management platform enable verified patient matching and chain-of-custody documentation that CGTs require. Because these therapies are living biological materials, there is no tolerance for tracking gaps. 3PLs that cannot provide integrated COI orchestration and verifiable cryogenic monitoring will find themselves excluded from this fast-growing segment.
Trend 5: Regulatory-Driven Serialization, Traceability, and Compliance Technology
The U.S. Drug Supply Chain Security Act (DSCSA) mandates interoperable, electronic tracing of products at the package level. The FDA established a 1-year stabilization period that ended on November 27, 2024. With full enforcement now active, the industry's primary challenge has shifted to "exception handling"—managing clerical errors and data mismatches where serialized product data doesn't match the physical shipment, causing severe operational disruptions at pharmacies and distributors.
Global serialization mandates are tightening:
Starting July 1, 2025, designated medical institutions in China must scan drug traceability codes before settling reimbursements through the national medical insurance system, with full compliance required by January 1, 2026. Non-compliance can result in administrative fines, suspension of product registration, and import bans. In the EU, the Falsified Medicines Directive (FMD) requires a unique identifier (2D DataMatrix) and an anti-tampering device on outer packaging, with maximum fines for falsifying medicines ranging up to €1 million in Spain.
Blockchain moves from pilot to production:
The FDA DSCSA Pilot Project demonstrated that blockchain could reduce the time required to identify affected products during a recall from three days to as little as ten seconds. Moving beyond pilots, the MediLedger network (administered by Chronicled) is now an operational blockchain network used by major players including Pfizer, McKesson, and AmerisourceBergen for DSCSA compliance, returns verification, and complex commercial contract alignment.
As enforcement tightens, companies are prioritizing logistics partners with established compliance infrastructure. VuRoyal's supply chain model — backed by drug business quality management standard certification, customs declaration unit registration, and third-party compliance solutions — is built to meet these requirements, helping clients avoid costly delays or enforcement actions.
What's Driving These Trends
These five trends share a common root: industry growth, regulatory pressure, and technology maturity are colliding at the same moment, raising the bar for what biopharma logistics must deliver.
Expanding Biologics and Vaccine Pipeline
The global biologics market reached $400.02 billion in 2024 and is on track to hit $653.34 billion by 2030 — an 8.5% CAGR driven by rising rates of cancer, autoimmune disorders, and genetic disease. This pipeline expansion is generating demand for logistics infrastructure that traditional pharma supply chains were never built to support.
The scale of that demand is visible in the numbers:
- 244 unique mRNA vaccine and therapeutic candidates in clinical development (as of March 2025)
- 48 FDA-approved cellular and gene therapy products currently on the market
- 3,243 ongoing clinical trials investigating gene, cell, or RNA therapies
- Monoclonal antibodies, CGTs, and novel adjuvants (such as saponin-based QS-21 and Matrix-M™) each require specialized cold chain and regulatory handling

Each of these modalities demands purpose-built logistics — not adaptations of conventional distribution.
Technology Enabling Real-Time Control
Technology advancement as an enabler: AI, IoT, and automation tools are now commercially available and scalable, enabling pharma logistics providers to operationalize real-time monitoring, predictive risk management, and digital compliance documentation at a level not previously possible. These technologies shift supply chain management from firefighting after failures to catching risks before they materialize.
Regulatory Intensification
Regulatory intensification: Global regulators are raising the bar on traceability, product integrity, and import/export compliance. These mandates are not optional — companies must either invest internally or partner with specialized distributors who already hold the certifications, licenses, and systems required. DSCSA is a clear case in point: what began as an IT implementation project has become a continuous operational function focused on exception handling and real-time verification.
Geopolitical and Cost Pressures
Geopolitical and cost pressures: Tariff volatility, trade restrictions, and pandemic-era supply disruptions have elevated supply chain resilience to a board-level priority. Companies are willing to pay a premium for logistics partners that offer proven reliability, regulatory compliance, and single-source accountability. The BIOSECURE Act and similar legislation are accelerating the structural shift away from concentrated API sourcing toward diversified, allied-nation supply networks.
How These Trends Are Impacting the Industry
These trends are producing measurable, interconnected changes across operations, business strategy, and workforce capabilities in the biopharma logistics sector.
Operational Impact
Cold chain requirements, serialization mandates, and CGT logistics complexity are reshaping warehouse design, fleet capabilities, and quality management workflows. Companies that cannot adapt are experiencing higher product loss rates and regulatory non-compliance penalties.
The shift from passive temperature logging to active IoT-enabled intervention requires new infrastructure investments. CGT logistics, in particular, demand dedicated cryogenic storage and chain-of-identity tracking systems unlike traditional bulk pharmaceutical distribution.
Business Impact
Biopharma companies are shifting procurement and distribution strategies—moving away from lowest-cost logistics models toward value-based partnerships with certified, compliant specialists. Specialty drugs now represent more than half of total pharmaceutical spending in the United States.
These high-cost therapies demand unique handling, storage, and administration requirements. Manufacturers now prioritize distributor competence, consistency, and reach: specifically, the ability to ensure product availability and reliably serve the intended patient population.
Top-performing procurement organizations ("Digital Masters") are allocating up to 24% of their budgets to technology. These investments in AI and digital tools are critical for managing increasing supply market complexity, driving resilience, and ensuring compliance with stringent cold chain and serialization mandates.
Workforce Impact
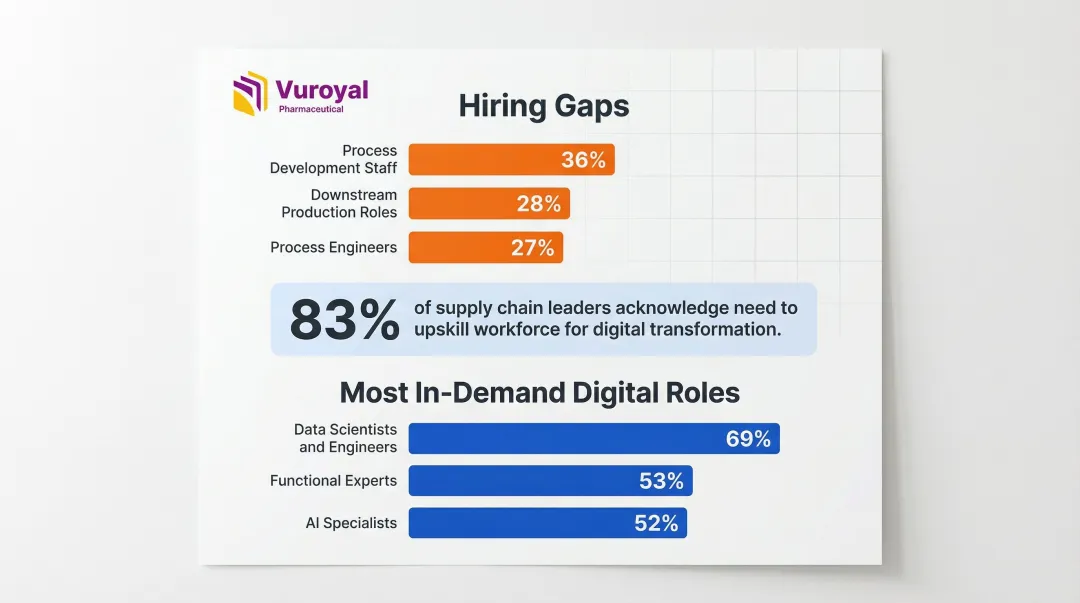
The complexity of modern biopharma logistics is creating demand for a new skills profile—professionals who combine regulatory knowledge, digital platform proficiency, and specialized cold chain expertise. According to BioPlan Associates' 2025 data:
- 36% of facilities cannot hire process development staff
- 28% struggle to fill downstream production roles
- 27% lack sufficient process engineers
- The average time-to-hire in Pharma & Life Sciences is 44 days
These hiring gaps extend beyond technical roles. 83% of biopharma supply chain leaders acknowledge the need to upskill or reskill a significant portion of their workforce for digital transformation. The most in-demand roles are data scientists and engineers (69%), functional experts (53%), and AI specialists (52%). Talent shortages in sustainability roles affect 34% of executives as well.
To offset these shortages, companies are directing R&D investment toward advanced automation—boosting efficiency while reducing dependence on scarce labor. Global pharma leaders are also expanding Global Capability Centers (GCCs) in talent-rich regions like India to handle data science, regulatory operations, and supply chain analytics.
Future Signals for Bio-Pharma Logistics
While the trends above are already visible, several early-stage signals point to how the biopharma logistics sector will evolve over the next one to three years. Companies that begin preparing now will gain a competitive head start.
Drone-Assisted Last-Mile Delivery for Biologics
Drone delivery for temperature-sensitive biologics is moving from pilot to scalable operations. In February 2026, Zipline signed an expansion agreement to provide full nationwide autonomous logistics coverage in Rwanda—the first country in Africa with an urban drone delivery network. In the U.S., UPS Flight Forward completed the first drone delivery of COVID-19 vaccines using custom cold chain packaging to maintain 2–8°C. A Michigan pilot further validated the approach, confirming that drones can hold payload temperatures averaging 17.3°C, well within acceptable ranges for common laboratory specimens.
Expanding Regulatory Reciprocity Between Major Markets
The U.S.-EU Mutual Recognition Agreement (MRA) allows drug inspectors to rely on inspection data collected across each other's borders. As of October 1, 2025, the EMA now voluntarily accepts FDA inspections of manufacturing sites outside the U.S. when reviewing human medicinal product applications.
The practical result: less duplicated inspection activity, faster cross-border approvals, and regulators redirecting attention toward higher-risk sites.
Blockchain Chain-of-Custody Platforms Reaching Operational Scale
The MediLedger network shows blockchain has moved well past proof-of-concept. It now supports DSCSA compliance, returns verification, and contract management across major pharmaceutical manufacturers and distributors—signaling that adoption is shifting from early experiments to industry standard.
Sustainability Metrics Entering Procurement Decisions
Large pharma companies are embedding emissions requirements into their supplier contracts. Pfizer's Scope 3 (value chain) GHG emissions run roughly four times higher than its direct operations—pushing the company to require that 65% of suppliers by spend commit to science-based emission reduction targets. Novartis has set similar expectations around waste reduction and eco-friendly materials.
For logistics providers, the implication is direct:
- Deploy electric vehicle fleets for last-mile and regional distribution
- Shift to reusable cold chain packaging to meet enterprise contract requirements
As cross-border pharmaceutical trade grows—especially between the U.S. and markets like China—organizations that already have certified import/export infrastructure, customs clearance capabilities, and regulatory guidance services in place will be positioned to capitalize on these opportunities without costly delays. VuRoyal's supply chain model—built on FDA registration, drug business licenses, quality management certifications, and customs declaration registration—is designed precisely for this environment, offering clinical trial sponsors and pharmaceutical manufacturers a single, compliant channel into the China market.
Conclusion
The forces reshaping bio-pharma logistics — cold chain innovation, AI-driven visibility, supply chain diversification, CGT complexity, and tightening regulatory traceability — are collectively redefining how pharmaceutical products move from manufacturer to patient. The market's projected growth to $234.58 billion by 2031 reflects not just expansion but fundamental transformation.
Manufacturers, clinical sponsors, and distributors that invest in strategic logistics partnerships now will be better positioned to protect product integrity, maintain compliance, and deliver life-saving therapies at scale. The $35 billion annual cost of cold chain failures and accelerating regulatory enforcement leave no room for reactive planning.
Organizations navigating this environment need partners with specialized cold chain capabilities, digital traceability, and built-in regulatory infrastructure. VuRoyal Pharmaceutical's combination of FDA-registered distribution, cold chain pharmaceutical transportation, and free regulatory guidance for drug registration reflects exactly the kind of integrated support this market demands.
Frequently Asked Questions
What is the biopharma supply chain?
The biopharma supply chain covers every step from sourcing raw materials—including Active Pharmaceutical Ingredients (APIs)—through manufacturing, to distributing finished products via compliant logistics networks. End users include hospitals, pharmacies, clinical trial sites, and research organizations.
What are the risks of the pharmaceutical supply chain?
Key risks span operational, regulatory, and geopolitical dimensions:
- Cold chain failures that degrade temperature-sensitive products
- API sourcing concentration in single geographies (notably China and India)
- Regulatory non-compliance penalties and counterfeit product infiltration
- Cybersecurity threats to digital tracking systems
- Geopolitical disruptions affecting cross-border trade
What is driving growth in the biopharma logistics market?
Growth is driven by the expanding biologics and vaccine pipeline (projected to reach $653.34 billion by 2030), increasing regulatory complexity including DSCSA serialization mandates, the rise of cell and gene therapies requiring specialized ultra-cold handling, and technology-driven demand for real-time supply chain visibility and compliance documentation.
How does cold chain logistics affect biopharma distribution?
Approximately 50% of new medicines require strict temperature control throughout transit and storage. Cold chain failures can compromise product efficacy, trigger regulatory rejection, or cause significant financial losses. These failures contribute to the industry's $35 billion annual spoilage crisis, making cold chain infrastructure a critical investment for manufacturers and distributors.
What role does AI play in biopharma supply chain management?
AI supports demand forecasting, predictive risk identification, real-time exception alerting, and automated compliance documentation. Notably, 69% of pharmaceutical companies have implemented AI-driven alerts for cold chain monitoring, and 40% cite demand forecasting as a top application—both reducing manual error and accelerating decision-making.
How can pharmaceutical companies reduce supply chain risk?
A resilient supply chain strategy combines people, process, and technology:
- Adopt dual-sourcing strategies to reduce geographic concentration
- Establish rigorous supplier qualification programs
- Invest in real-time tracking and IoT monitoring technology
- Partner with certified logistics specialists who offer full supply chain accountability and proven cold chain infrastructure


