
Introduction
A single temperature excursion during transit can render a life-saving drug completely ineffective. For biologics, cell and gene therapies, and temperature-sensitive vaccines, the margin for error in distribution is essentially zero.
Even brief deviations can turn a $2.125 million gene therapy dose into medical waste—or convert a vaccine shipment into a public health liability.
Cold chain is often treated as background infrastructure, but its real value and real risk shows up at the point of patient care, regulatory audit, and product loss accounting. The biopharma industry loses approximately $35 billion annually due to temperature-controlled logistics failures, with single-dose losses for advanced therapies reaching catastrophic levels.
This article breaks down why cold chain logistics is a core operational priority in pharma: what's at stake when it fails, what good practice actually requires, and why the industry can no longer treat it as a back-office function.
TLDR
- Cold chain logistics maintains continuous, validated temperature control across the entire pharmaceutical supply chain—from manufacturer to patient
- Temperature-sensitive products like vaccines, biologics, and insulin lose efficacy or become harmful after brief temperature excursions
- Effective cold chain protects patient safety and prevents costly product losses from temperature excursions
- FDA and WHO have established strict Good Distribution Practice (GDP) requirements for cold chain operations
- For manufacturers and clinical trial sponsors, choosing a compliant logistics partner is the most consequential supply chain decision
What Is Cold Chain Logistics in Pharma?
Cold chain logistics is a temperature-controlled supply chain system designed to maintain pharmaceutical products within their required temperature ranges—from production through storage and transit to the final point of care.
Temperature Bands in Pharmaceutical Distribution
Which temperature band applies depends on the drug's formulation and stability profile:
- Controlled Room Temperature: 15–25°C, with excursions between 15°C and 30°C allowed per USP standards
- Refrigerated: 2–8°C for monoclonal antibodies, unopened insulin, and traditional vaccines
- Frozen: −10°C to −25°C (Moderna's mRNA-1273, for example, requires −50°C to −15°C)
- Ultra-cold: Below −70°C for certain mRNA vaccines like Pfizer-BioNTech's original formulation and specific gene therapies

Every step in this chain is a patient safety checkpoint — ensuring that what leaves the manufacturer arrives pharmacologically intact.
Key Advantages of Cold Chain Logistics in Pharma
The advantages below are grounded in operational and business outcomes, with direct implications for product quality, regulatory standing, financial performance, and patient safety.
Advantage 1: Preserving Product Integrity and Protecting Patient Safety
The primary function of cold chain logistics is to ensure that temperature-sensitive pharmaceutical products—vaccines, biologics, monoclonal antibodies, insulin, oncology treatments—maintain their chemical stability and therapeutic efficacy from origin to administration.
How Cold Chain Systems Protect Product Integrity:
Cold chain systems deliver product integrity through refrigerated storage facilities, validated transport packaging (insulated shippers, gel packs, dry ice, phase-change materials), and continuous temperature monitoring devices (dataloggers and real-time IoT sensors) that detect and flag excursions before delivery.
The Cost of a Single Excursion:
A temperature excursion doesn't just degrade the product. It can make a drug appear effective while delivering a subtherapeutic dose, posing direct patient harm. The WHO estimates that over half of vaccines are wasted globally each year due to temperature control failures, logistics challenges, and shipment-related issues.
Patient harm from out-of-spec medication creates medical liability, erodes institutional trust, and can trigger product recalls affecting entire batches. For high-cost biologics and oncology treatments, even a single shipment loss can represent hundreds of thousands of dollars in wasted product:
- Zolgensma (spinal muscular atrophy): $2,125,000 per treatment
- Luxturna (inherited retinal dystrophy): $850,000 per patient
- Kymriah (B-cell acute lymphoblastic leukemia): $475,000 per treatment
- Yescarta (large B-cell lymphoma): $373,000–$399,000 per treatment
KPIs Impacted:
- Product rejection rate
- Temperature excursion frequency
- Shipment acceptance rate
- Patient adverse event rate tied to drug administration
When This Advantage Matters Most:
This advantage is especially critical for biologics, cell-based therapies, mRNA vaccines, and clinical trial investigational drugs—where temperature sensitivity is highest and where there is zero tolerance for compromise.
Advantage 2: Regulatory Compliance and Audit Readiness
Pharmaceutical cold chain operations are subject to rigorous regulatory oversight from bodies including the FDA, EMA, and WHO. Good Distribution Practice (GDP) guidelines mandate validated temperature monitoring systems, accurate record-keeping, and documented corrective actions for any excursions.
How a Well-Executed Cold Chain Creates Compliance Infrastructure:
In real-world operations, calibrated monitoring devices generate tamper-proof temperature logs, SOPs govern response protocols, thermal mapping validates storage environments, and all data is stored in audit-ready formats.
What Non-Compliance Actually Costs:
Companies with thorough cold chain documentation enter regulatory audits from a position of confidence. They can demonstrate continuous control, traceability, and accountability across every stage of the supply chain.
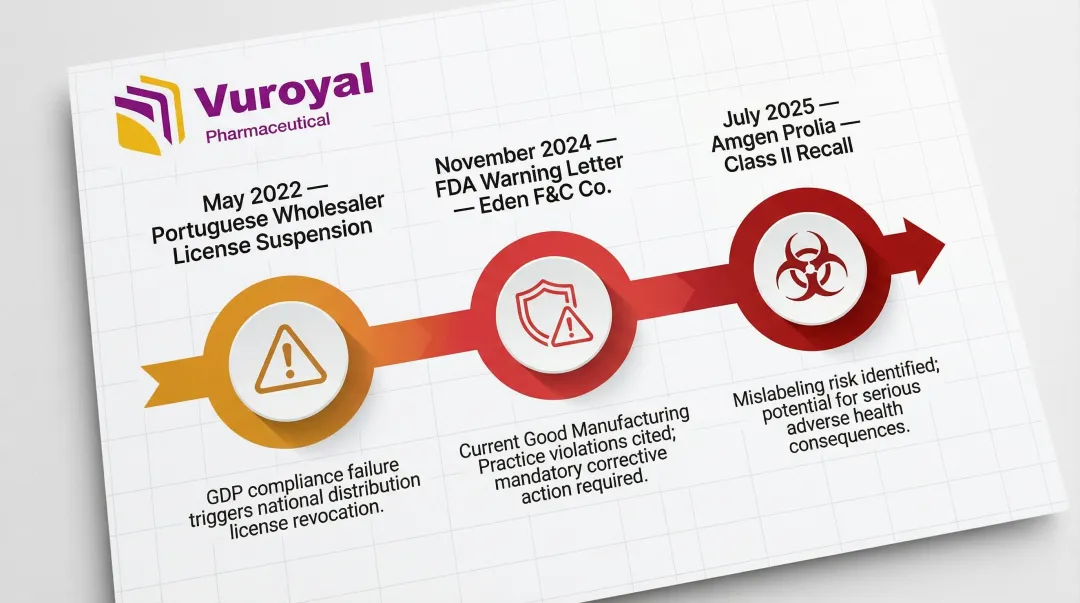
Recent regulatory enforcement actions illustrate the consequences of non-compliance:
- July 2025: Class II recall of Amgen's Prolia due to transit temperature excursions
- May 2022: Portuguese wholesaler's license suspended after failing to trigger emergency procedures during cold chamber breakdown
- November 2024: FDA Warning Letter to Eden F&C Co. for failing to ensure refrigerated truck ambient temperature remained below 3.3°C with continuous monitoring
Pharmaceutical importers and distributors operating across borders, such as between the US and Asia, face multi-jurisdictional regulatory requirements. A compliant cold chain partner who understands both FDA and destination-country regulations reduces that exposure significantly.
In clinical trials specifically, cold chain documentation is part of the trial's quality evidence package. Failures here can invalidate trial data entirely.
KPIs Impacted:
- Audit pass rate
- Number of GDP deviations
- Documentation completeness score
- Regulatory response time for excursion incidents
When This Advantage Matters Most:
Compliance pressure is highest during international shipments, cross-border customs clearance, and for investigational new drugs in clinical trials, where regulatory scrutiny is most intense.
Advantage 3: Financial Risk Mitigation and Supply Chain Reliability
Cold chain failures generate compounding financial losses across wasted product, replacement costs, logistics rerouting, regulatory penalties, recall expenses, and reputational damage. A single temperature breach can trigger all of them.
How a Well-Managed Cold Chain Prevents This:
Through proactive real-time alerts that allow in-transit intervention before a shipment is compromised, contingency planning (backup carriers, redundant monitoring), and risk-mapped routing that accounts for seasonal weather and infrastructure reliability.
The Financial Case for Cold Chain Investment:
The biopharma cold chain third-party logistics market was valued at $30.59 billion in 2024 and is projected to reach $74.46 billion by 2033, a 10.54% CAGR. That trajectory reflects how seriously the industry now treats cold chain as a core business function.
For pharmaceutical manufacturers and distributors, an unreliable cold chain creates downstream shortages. Hospitals and clinics cannot dispense medications on schedule, which disrupts patient care and strains provider relationships. A 2019 Peli BioThermal survey revealed that 44.6% of biopharma companies report multiple temperature excursions per year, with 16% experiencing them monthly.
For specialty drugs, biologics, and vaccine adjuvants moving through international supply chains, a single failed shipment can delay a clinical program or leave a market undersupplied. The consequences compound quickly.
KPIs Impacted:
- Cost per temperature excursion incident
- Product waste as a percentage of shipment value
- On-time delivery rate
- Supply continuity index
When This Advantage Matters Most:
Financial risk is highest in long-distance, multi-leg, or cross-border shipments where handoff points between carriers or facilities create the greatest temperature vulnerability, and where real-time monitoring and experienced logistics partners provide the most protection.
What Happens When Cold Chain Logistics Is Ignored or Undermanaged
Organizations that treat cold chain as a cost to minimize — rather than a system to invest in — face a predictable chain of serious consequences:
Compromised product quality. Direct adverse events are rare (only 7% of VAERS reports noted AEs, mostly local reactions), but indirect costs are not. Subtherapeutic doses appear clinically administered yet fail patients silently. A single unreported cold chain breach at an Australian clinic forced the recall and revaccination of 1,600 patients.
Regulatory violations and import holds. Inadequate temperature records can halt distribution operations and jeopardize operating licenses. In 2023, the FDA issued a Warning Letter and triggered a nationwide recall for Claire Ellen Products after discovering the firm failed to monitor storage conditions — including temperature and humidity — for heat-sensitive drugs.
Significant financial losses. Product waste, emergency re-shipments, and insurance claims compound quickly as pipelines grow more specialized. Supply chain delays for specialty biologics can cost up to $600,000 per day; delays for blockbuster drugs can reach up to $8 million per day.
Erosion of trust. Healthcare providers, clinical trial sponsors, and institutional buyers rely on supply chain reliability for patient care continuity. That trust is hard to rebuild — and the data shows how easily it breaks. Systematic reviews reveal that 75% to 100% of vaccine shipments in longitudinal studies experience freezing temperatures, often from poorly conditioned ice packs.
How to Get the Most Value from Pharmaceutical Cold Chain Logistics
Cold chain effectiveness is an ongoing operational discipline. It pays off when applied consistently and reviewed systematically — not just during audits or incident reports.
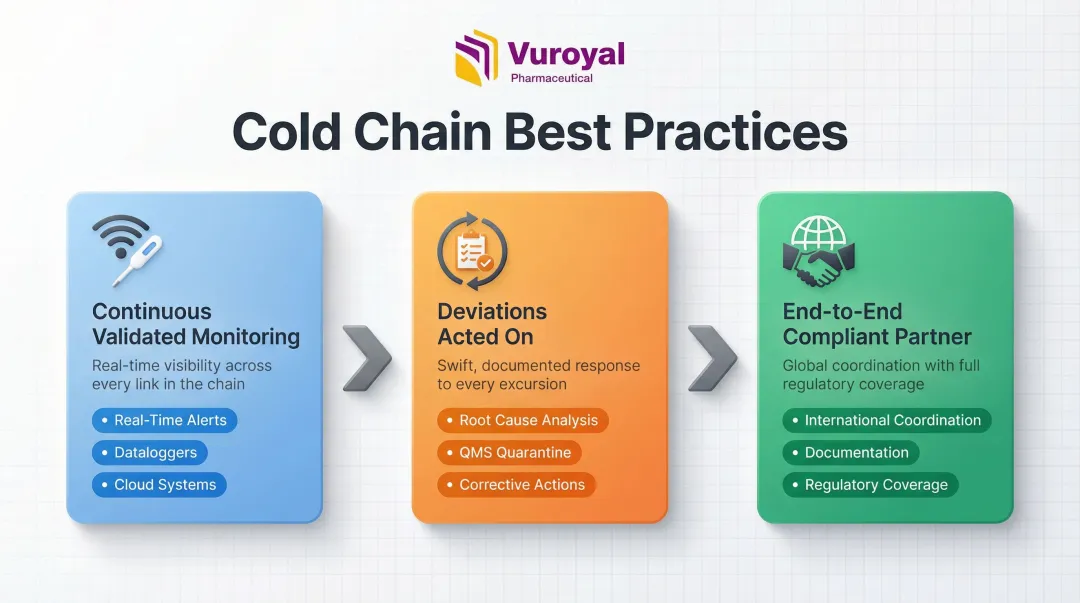
Three practices consistently separate high-performing cold chains from those that merely pass audits:
- Continuous, validated monitoring — Real-time IoT devices, calibrated dataloggers, and cloud-based alert systems should cover primary transport, last-mile delivery, and storage — not just the segments that get inspected. Specialized clinical trial couriers with dedicated cold storage networks achieve <0.3% shipment failure rates, compared to the 2–5% industry standard.
- Deviations acted on, not just logged — Every excursion should trigger root cause analysis and corrective action. Automated Quality Management Systems (QMS) can quarantine affected products and initiate Corrective and Preventive Actions (CAPAs) the moment a temperature breach is detected.
- **End-to-end coordination by a compliant logistics partner** — This matters most for international shipments crossing multiple regulatory jurisdictions, where documentation gaps and customs delays create the highest risk of cold chain failure.
VuRoyal Pharmaceutical provides specialty drug logistics with cold chain transportation services for pharmaceutical shipments to and from the U.S. and China. As an FDA-registered wholesaler with customs declaration qualifications, VuRoyal supports research organizations, clinical trial sponsors, and pharmaceutical manufacturers with compliant, end-to-end distribution.
Conclusion
Cold chain logistics ultimately determines whether a pharmaceutical product reaches a patient in the same condition it left the manufacturer — any gap in temperature control, handling, or documentation puts both efficacy and safety at risk.
When cold chain management is treated as a core operational discipline rather than a shipping formality, the returns are concrete: fewer product losses, faster regulatory clearance, and supply chains that hold up under audit. For distributors handling reference drugs, vaccine adjuvants, and clinical trial materials, that discipline is built into every shipment.
In a pharmaceutical environment defined by rising product complexity and tightening global regulation, cold chain rigor is simply the cost of operating responsibly — and the clearest indicator of whether a distribution partner can be trusted with what matters most.
Frequently Asked Questions
What is the cold chain in pharma?
The cold chain in pharma is the end-to-end temperature-controlled system used to store, transport, and distribute temperature-sensitive pharmaceutical products—including vaccines, biologics, and insulin—within their required temperature ranges, from manufacturer to patient.
What are the 7 C's of logistics?
The 7 C's of logistics are: the right Product, Condition, Quantity, Place, Time, Customer, and Cost. In cold chain pharma logistics, Condition is the most critical C. Temperature integrity determines whether a product is safe and effective when it reaches the patient.
What temperature ranges are used in pharmaceutical cold chain logistics?
The main temperature bands are CRT (15–25°C), refrigerated (2–8°C), frozen (−10 to −25°C), and ultra-cold (below −70°C for certain mRNA vaccines). The required range is determined by the specific drug's stability profile, and FDA label revisions based on updated stability data can authorize broader storage conditions for certain products.
What happens if the pharmaceutical cold chain is broken?
A cold chain break—or temperature excursion—can cause pharmaceutical products to degrade, lose efficacy, or become harmful, requiring the affected shipment to be discarded. The consequences extend to patient safety risks, regulatory violations, and significant financial losses.
How do pharmaceutical companies ensure cold chain compliance with FDA requirements?
Compliance is maintained through validated temperature monitoring systems, documented SOPs, calibrated dataloggers or real-time IoT sensors, thermal mapping of storage facilities, and audit-ready temperature records. All practices must align with FDA regulations and GDP guidelines.
What is Good Distribution Practice (GDP) in pharma logistics?
GDP is a quality standard established by regulatory bodies that defines how pharmaceutical products must be stored, transported, and distributed to maintain their integrity—covering temperature monitoring, documentation, personnel training, and corrective action protocols throughout the supply chain.


