
Introduction
Biopharma supply chains have never faced pressure like this. Drug shortages hit 323 active disruptions in Q1 2024, the highest level on record. Geopolitical tensions are forcing a fundamental reassessment of sourcing strategies, particularly as the BIOSECURE Act reshapes access to Chinese contract manufacturers. Regulatory pressure is compounding the problem: warning letters to drug manufacturers surged 50% in FY2025, with FDA heavily targeting inadequate sponsor oversight of CMOs and supplier qualification failures.
For research organizations, clinical trial sponsors, and pharmaceutical manufacturers, these pressures translate directly to delayed treatments, halted trials, and patient harm. Generic sterile injectables represent 67% of all shortages, hitting hospital care and cancer treatment hardest.
The pandemic made one thing clear: seemingly minor components — glass vials, rubber stoppers, backing paper — can halt entire production lines when single-source dependencies fail.
This article provides a strategic roadmap for navigating these challenges. You'll understand the structural imperatives reshaping biopharma supply chain management and walk away with concrete best practices to build resilience and maintain uninterrupted access to critical pharmaceutical substances.
Key Takeaways
- Record shortages, geopolitical risk, and API concentration demand proactive supply chain strategy
- Safety stock is now 35% above 2019 baselines — supplier diversification is no longer optional
- FDA holds sponsors strictly liable under 21 CFR 211.22; compliance and supplier qualification are inseparable
- Temperature excursions cost the industry $35 billion annually — cold chain infrastructure is non-negotiable
- Early AI adopters are cutting forecast error by 25%, shifting supply chains from reactive to predictive
The Current State of Biopharma Supply Chains: Why the Stakes Have Never Been Higher
Record Drug Shortages and Structural Vulnerabilities
The American Society of Health-System Pharmacists tracked 216 active drug shortages in early 2026, down from the all-time high of 323 in Q1 2024 but still critically elevated. Seventy-five percent of active shortages began in 2022 or later, indicating persistent rather than transient supply chain stress. Generic sterile injectables—critical for hospital care and cancer treatment—remain the most vulnerable category.
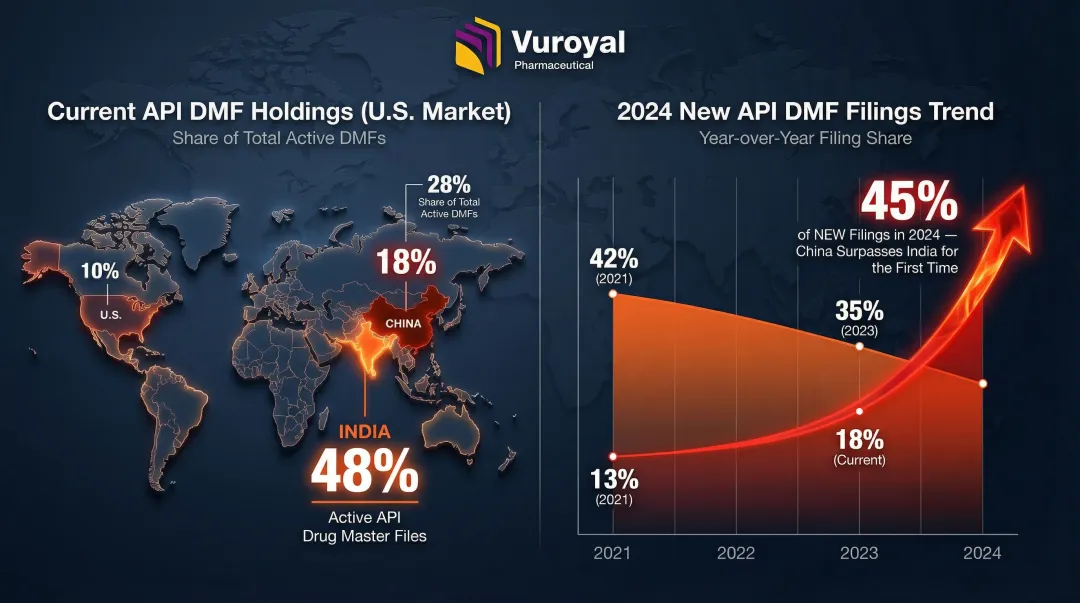
The structural problem runs deeper than headline numbers suggest. India accounts for 48% of active API Drug Master Files for the U.S. market, while China holds 18%, up from 13% in 2021. More concerning: China captured 45% of new API DMF filings in 2024, surpassing India for the first time. This geographic concentration creates single-point-of-failure dynamics — ones COVID-19 made impossible to ignore.
The BIOSECURE Act: Immediate Strategic Implications
The BIOSECURE Act became law on December 18, 2025, as Section 851 of the FY2026 National Defense Authorization Act. Unlike earlier drafts, the enacted legislation doesn't name specific entities—instead, it establishes a dynamic designation process led by the Office of Management and Budget (OMB), which must publish an initial list of "biotechnology companies of concern" by December 18, 2026.
Key compliance timelines:
- Prohibitions take effect 60 days (for DoD 1260H-listed entities) or 90 days (for OMB-designated entities) after Federal Acquisition Regulation revisions
- Legacy contracts receive a five-year grandfathering period
- New contracts with designated entities will be restricted within 60-90 days of FAR publication
Companies are already re-evaluating sourcing strategies regardless of the act's final implementation timeline. The strategic imperative is immediate: map tier-1 through tier-3 suppliers to identify exposure to Chinese CDMOs and initiate technology transfers to alternative geographies.
Beyond APIs: The Hidden Threat of Consumables
COVID-19 taught the industry an expensive lesson about overlooked dependencies. Shortages of container closure system components—specifically glass vials and stoppers—became severely constrained as manufacturers responded to vaccine demand. State officials reported severe shortages of specialized testing supplies, reagents, and plastic consumables like pipettes.
These aren't high-tech components. Yet single-source dependencies in backing paper, borosilicate vials, or specific chemical reagents can halt production just as effectively as API shortages. Two figures capture the exposure:
- Only 6% of companies report full end-to-end supply chain visibility
- Half of all supply chain disruptions originate at tier-2 and tier-3 suppliers
From Reactive to Predictive: The Strategic Shift
Most biopharma companies still learn about disruptions after they happen. Yet traditional supply chain monitoring focuses almost exclusively on tier-1 relationships — the suppliers you know — while leaving the deeper tiers unmonitored.
Closing that gap is less a technology problem than an organizational one. Companies that build cross-functional visibility across suppliers, logistics, and demand signals can forecast disruptions and act before shortages materialize.
Strategic Imperative 1: Diversify Supply Sources and Build Redundancy
The Foundational Imperative: Multi-Source, Multi-Geography Strategy
Supply source diversification is no longer contingency planning—it's the baseline requirement for pharmaceutical operations. Companies maintaining multiple qualified suppliers across different geographies for critical APIs, excipients, and raw materials are simply better positioned to absorb disruption than those that don't.
"China + 1" sourcing options by geography:
- India – Clinical trial share growth up 109% since 2019; strong API manufacturing capacity but still relies on China for 70% of bulk drug imports
- Southeast Asia – Vietnam and Indonesia seeing significant FDI increases; emerging manufacturing hubs with cost competitiveness
- European Union – Higher quality standards and regulatory alignment; significantly higher unit costs and longer qualification timelines
- United States – Maximum supply predictability; cost-prohibitive for full reshoring of generics
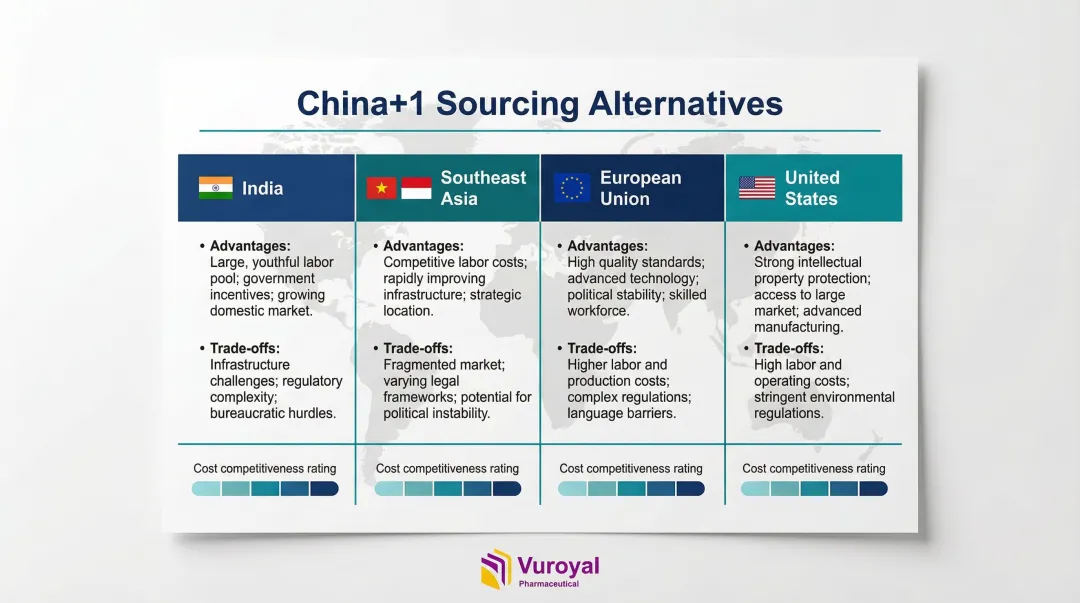
Each geography presents distinct cost-capacity trade-offs. API manufacturing costs in China run 35-40% lower than U.S. production, making complete reshoring economically unfeasible for most generic drugs.
The Reshoring Reality: Hybrid Strategies Offer Practical Paths
Full reshoring faces significant barriers: labor costs, environmental regulations, capital requirements, and idle manufacturing capacity. Friend-shoring has emerged as a pragmatic response, aligning economic resilience with strategic diplomacy by creating redundant, geographically diversified nodes in friendly jurisdictions.
In practice, most companies are landing on hybrid models. Common configurations include:
- Domestic production for critical or shortage-prone drugs, international sourcing for generics
- Regional hubs in allied nations for specific therapeutic categories
- Dual-qualified suppliers in two or more geographies for highest-risk APIs
From Just-in-Time to Just-in-Case: The Buffer Stock Reality
Pharmaceutical firms maintained safety stock buffers 35.2% above 2019 baselines through 2023, the most persistent buffer increases across industries. This resilience premium reflects ongoing regulatory complexity and critical dependencies in biologics supply chains, where single-source constraints remain endemic.
Strategic buffer stock considerations:
- Identify high-risk, single-source items requiring dedicated safety stock
- Balance buffer inventory against working capital constraints
- Implement AI-driven multi-echelon inventory optimization to dynamically adjust stock levels based on real-time demand signals
- Revisit lean inventory models for critical materials post-pandemic
Map the Entire Supply Chain: Beyond Tier-1 Visibility
Half of all supply chain disruptions stem from tier-2 and tier-3 suppliers, yet only 12% of firms track disruption at tier-3 and beyond. Many disruptions originate at levels most companies don't monitor—a backing paper supplier, a raw material intermediary, a specialty chemical manufacturer.
Action steps:
- Deploy digital supply network mapping tools to illuminate sub-tier dependencies
- Qualify secondary suppliers for critical packaging and consumable components
- Establish monitoring protocols for tier-2 and tier-3 financial health and performance
- Build redundancy at multiple supply chain levels, not just tier-1
| Sourcing Strategy | Primary Geography | Key Advantages | Trade-offs & Barriers |
|---|---|---|---|
| China-Centric | China | 35-40% lower API costs; massive scale | High geopolitical risk; single-point-of-failure vulnerability |
| China+1 | India, Southeast Asia | Diversifies risk; maintains cost competitiveness | India relies on China for 70% of bulk drug imports |
| Friendshoring | EU, Abraham Accords Nations | Strategic alignment; high quality standards | Higher unit costs; significant technology transfer time |
| Reshoring | United States | Maximum predictability; regulatory compliance | Cost-prohibitive for generics; workforce and capital barriers |
Strategic Imperative 2: Supplier Qualification, Compliance, and Relationship Management
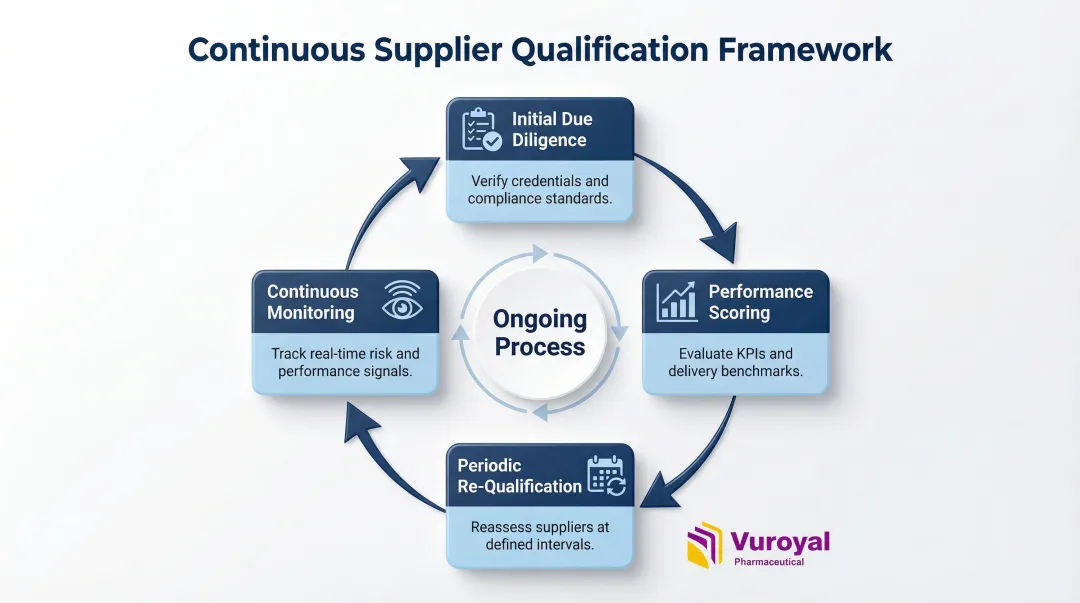
Continuous Qualification: Not a One-Time Event
Supplier qualification in biopharma is a continuous process, not a checkbox exercise. Certifications must stay current, GMP compliance must be verified, and performance must be tracked over time. The FDA and EMA require traceability and documentation on demand—under 21 CFR 211.22, the quality control unit has responsibility to approve or reject all components.
Robust supplier qualification framework components:
- Initial due diligence – Certifications, audit history, financial health, GMP compliance verification
- Performance scoring – On-time delivery metrics, quality performance, corrective action responsiveness
- Periodic re-qualification – Scheduled audits, certification renewals, risk-based reassessment cadence
- Continuous monitoring – Real-time tracking of deviations, OOS results, regulatory actions
21 CFR 211.84 mandates that a Certificate of Analysis may be accepted from a supplier provided at least one specific identity test is conducted by the manufacturer and the supplier's reliability is established through validation. EMA GDP guidelines require appropriate qualification and approval of suppliers prior to procurement, with documented procedures governing procurement and mandatory periodic rechecks.
The Cost of Poor Oversight: Regulatory Enforcement Surge
FDA warning letters to drug manufacturers surged 50% in FY2025, heavily targeting sponsor oversight of contract manufacturing organizations and supplier qualification failures. The FDA holds sponsors strictly liable for CMO GMP non-compliance—inadequate oversight leads to import alerts, clinical trial delays, and product recalls.
Case study: Darmerica LLC (December 2025)
Darmerica was cited for critical failures in supplier qualification—the firm requalified an API supplier despite conflicting information about FDA inspection history and failure to provide required stability data. The FDA noted Darmerica imported and distributed drugs from that supplier despite apparent failure to comply with quality agreement terms.
Common warning letter citations:
- Requalifying suppliers without independent verification of certifications and audit history
- Accepting supplier test methods without formal validation documentation
- Failing to investigate customer complaints beyond initially reported lots
- Inadequate Quality Agreements with contract manufacturers
Escape "Spreadsheet Purgatory": Centralize Supplier Data
Forty percent of Chief Procurement Officers said their organizations don't define or measure relevant ESG factors, largely because procurement data is scattered across ERPs, contract repositories, spreadsheets, and supplier systems. That fragmentation creates compliance blind spots that auditors—and warning letters—tend to find first.
Centralized supplier information management priorities:
- Unified digital platforms replacing fragmented spreadsheets
- Single source of truth for supplier certifications, audit results, performance metrics
- Automated alerts for expiring certifications or quality events
- Integration with quality management systems for real-time deviation tracking
Performance Monitoring: Essential, Not Optional
Centralized data infrastructure is only useful if it feeds active monitoring. In biopharma, a late shipment or batch failure can derail a clinical trial or create a shortage of a life-critical drug—which means tracking supplier performance continuously, not just at qualification.
Core performance monitoring practices include:
- Tracking on-time delivery and batch acceptance rates against contracted SLAs
- Logging deviation frequency and time-to-CAPA resolution per supplier
- Scoring suppliers quarterly and flagging deteriorating trends before they become failures
- Requiring suppliers to notify you of any regulatory actions, inspections, or internal quality events
Choosing the right distribution partners is equally important. VuRoyal Pharmaceutical, an FDA-registered and Massachusetts-approved pharmaceutical wholesaler, structures its GDP Standard Operation Protocol around USP, 21 CFR, PDMA, and DSCSA requirements—using third-party compliance verification to maintain ongoing adherence to state and federal regulations.
Best Practices for Biopharma Supply Chain Operations
Demand-Driven Planning: Align Procurement with Clinical and Manufacturing Timelines
Biologics represent 49% of the global pharmaceutical pipeline and have driven much of the industry's growth over the last decade. Manufacturing biologics—from cell therapies to fermentation-based products—involves extended lead times and process complexity that traditional small-molecule planning simply doesn't account for. That makes demand-driven procurement a baseline requirement, not a best practice.
Core planning disciplines include:
- Aligning procurement cycles with clinical trial enrollment timelines and manufacturing schedules
- Forecasting projected patient demand rather than purchasing reactively
- Accounting for extended lead times in biologic drug and specialty material sourcing
- Building buffer stock for critical materials with long or uncertain lead times
Cross-Functional Procurement: Break Down Silos
Effective biopharma supply chain management requires active collaboration between procurement, R&D, regulatory affairs, quality assurance, and legal—not siloed purchasing decisions. High-performing procurement organizations invest in agility by developing talent and accessing capabilities through cross-functional collaboration. When procurement is integrated across functions, the downstream effects are significant:
- Supplier qualification accelerates when quality and regulatory teams are involved from the start
- Demand forecasting improves through direct R&D and manufacturing collaboration
- Compliance risk drops when legal reviews supplier agreements before execution
- Predictive analytics across functions enables faster, fact-based sourcing decisions
Strategic Spend Management and Supplier Consolidation
Spend management becomes strategic when it reflects both cost efficiency and supply chain resilience — not just the cheapest option at the moment of purchase.
Practical approaches include:
- Consolidating supplier relationships where category overlap exists to gain volume leverage
- Renegotiating contracts periodically based on performance data and market conditions
- Tracking spend by category and supplier to surface optimization opportunities
- Aligning purchasing decisions with long-term business goals, not short-term cost pressure
- Weighing cost efficiency against supply chain resilience at every sourcing decision
Clear SLAs and Documentation Standards
Once procurement and spend strategies are in place, operational reliability depends on what's formally agreed — and documented — with every supply chain partner.
Establish clear service level agreements and documentation standards with all supply chain partners, including:
- Defined escalation protocols for quality failures
- Response time requirements for delivery delays
- Regulatory non-conformance notification procedures
- Chain-of-custody documentation requirements
- Quality Agreement terms covering all critical processes
Cold Chain and Specialty Drug Logistics: A Non-Negotiable for Biologics
The $35 Billion Excursion Problem
The biopharma industry loses approximately $35 billion annually due to temperature-controlled logistics failures. A survey of industry leaders revealed 44.6% reported multiple temperature excursions per year, and 16% said excursions were monthly. Forty-one percent of excursions exceed four degrees, and 21% exceed eight degrees.

Beyond financial write-offs, temperature breaches may result in loss of potency or stability and pose severe patient safety risks through reduced efficacy or harmful degradation products.
Why Cold Chain is Uniquely Critical in Biopharma
Biologics, vaccines, and specialty compounds are highly sensitive to temperature excursions, vibration, and environmental factors. A compromised product may not be visually distinguishable from an intact one, creating invisible patient safety risk. Cold chain planning should begin at the development stage, not at commercial launch.
Compliant cold chain infrastructure requirements:
- Validated temperature-controlled packaging (active or passive systems)
- Continuous monitoring with data loggers or real-time sensors
- GDP-compliant transportation protocols
- Chain-of-custody documentation from manufacturer to end user
EMA GDP guidelines mandate that for temperature-sensitive products, qualified equipment must be used, and if temperature-controlled vehicles are used, temperature mapping under representative conditions must be carried out. WHO TRS 961 requires air temperature monitoring systems with minimum recording frequency of six times per hour.
Specialty Drug Logistics Providers: Reducing Compliance Burden
Specialty drug logistics providers reduce the compliance burden for research organizations and clinical trial sponsors importing specialty materials across borders. VuRoyal Pharmaceutical, for example, offers cold chain pharmaceutical transportation with end-to-end customs clearance capabilities, validated infrastructure, and regulatory expertise to manage cross-border pharmaceutical compliance.
The global pharmaceutical logistics market was estimated at $99.33 billion in 2024 and is projected to reach $208.26 billion by 2033, growing at 8.5% CAGR. The broader cold chain market is projected to reach $1,611 billion by 2033, growing at 20.5% CAGR.
| Packaging Type | Regulatory Compliance | Cost & Performance Profile |
|---|---|---|
| Active ULDs | Highly compliant; continuous data logging; minimal excursion risk | High cost; heavier weight affects fuel; strongest protection |
| Passive Packaging | Compliant if validated against seasonal thermal profiles (such as ISTA 7D) | Lower cost; susceptible to temperature swings and tarmac delays |
The Role of Digital Technology and AI in Supply Chain Resilience
From Reactive to Predictive: The AI Advantage
In a recent McKinsey survey, 78% of respondents reported their organizations use AI in at least one business function. High-performing organizations commit more than 20% of their digital budgets to AI technologies.
Organizations that invest in integrated data platforms gain the ability to anticipate disruptions—supplier financial stress, geopolitical events, raw material shortages—before they escalate. Agentic AI autonomously senses and mitigates supply chain risk, monitors tier-1 and tier-2 suppliers, and recommends alternative suppliers that balance risk and cost.
Digital Twins and AI Forecasting: Measurable ROI
Digital twins in action:
- Takeda achieved a 4.0% yield improvement using digital twin technology and machine learning at its Hikari site to optimize cooling water valve openings
- Pfizer leveraged GPU-native CFD software to build digital twins of microbioreactors, accelerating manufacturing innovation and providing a roadmap for process scale-up
AI-driven demand forecasting:
A proof-of-concept for a pharma distributor utilizing machine learning forecasting showed 25% forecast error reduction and 40% expected inventory excess reduction within six months, while securing higher service levels (>95% compared to previous 80%).

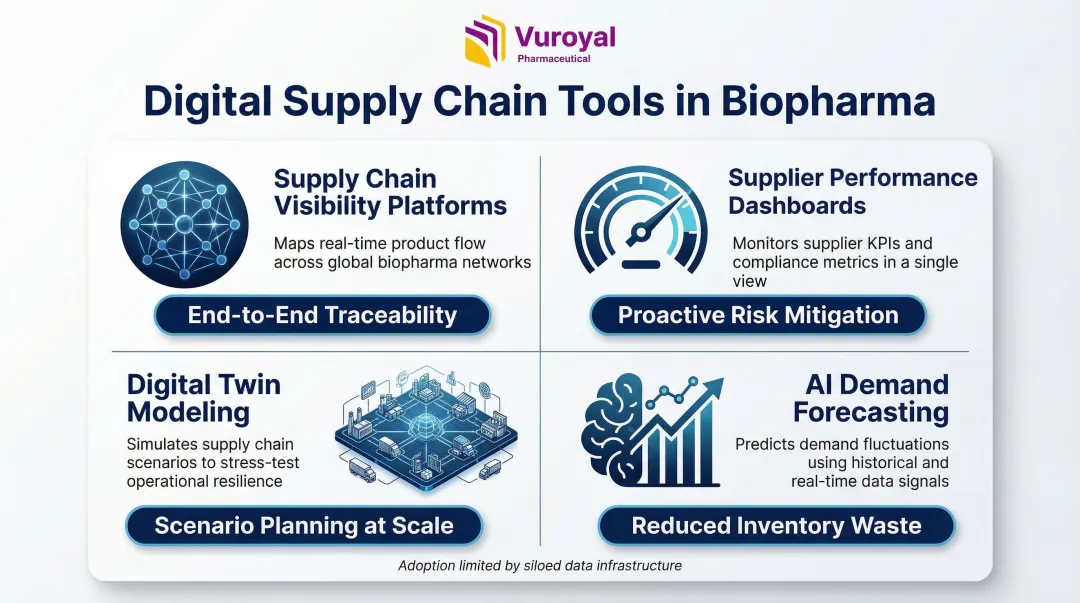
Practical Digital Tools Gaining Adoption
- Supply chain visibility platforms track shipments and supplier activity across multi-tier networks in real time
- Supplier performance dashboards centralize quality metrics, on-time delivery rates, and corrective action logs
- Digital twin modeling runs scenario planning for manufacturing bottlenecks and process constraints
- AI demand forecasting applies machine learning models enriched with external supply and market data
Most of these tools already exist and are proven. What slows adoption in biopharma is disconnected, siloed systems that block the enterprise-wide data integration these tools require to function effectively.
Public-Private Partnerships and Collective Intelligence
Two federal initiatives are actively reshaping how supply chain intelligence is gathered and shared across the industry:
- The FDA's Quality Management Maturity (QMM) program encourages manufacturers to adopt advanced quality practices; after two pilots between 2020 and 2022, the FDA began evaluating nine volunteer manufacturers in 2024
- The FY2026 NDAA directs HHS to coordinate public-private partnerships that map pharmaceutical supply chains and use data analytics to flag vulnerabilities posing national security risks
For biopharma distributors and clinical trial sponsors, these programs signal that API shortage early-warning systems and joint regulatory strategies are moving from aspiration to enforceable expectation.
| Maturity Stage | Data Management | Forecasting & Planning | Supplier Visibility |
|---|---|---|---|
| Legacy / Siloed | Fragmented spreadsheets; disconnected ERPs | Static historical averages; high forecast error | Tier-1 only; reactive disruption management |
| Integrated | Unified data layer; single source of truth | Machine learning models; dynamic inventory optimization | Digital supply network mapping; proactive risk alerts |
| Agentic AI | Autonomous data enrichment and synchronization | Digital twins simulating what-if scenarios and bottlenecks | Continuous sub-tier risk monitoring with automated mitigation recommendations |
Frequently Asked Questions
What are the biggest risks facing biopharma supply chains today?
The top risks include geographic concentration of API production in China and India, record-high drug shortages (particularly generic sterile injectables), regulatory and legislative pressures such as the BIOSECURE Act, and single-source dependencies across multiple supply chain tiers including tier-2 and tier-3 suppliers.
How does regulatory compliance affect biopharma supply chain strategy?
Regulatory requirements from FDA, EMA, and other agencies require all supply chain partners to maintain current certifications, GMP compliance, and full documentation traceability. Compliance is a structural requirement for supplier selection, with sponsors held strictly liable for CMO performance under 21 CFR 211.22.
What is cold chain management and why is it critical in biopharmaceutical distribution?
Cold chain management refers to temperature-controlled handling and transportation of biologics and other sensitive compounds from manufacturer to end user. Failure to maintain cold chain integrity can compromise product safety and patient outcomes, with the industry losing $35 billion annually to temperature excursions.
How can biopharma companies reduce dependence on single-source suppliers?
Implement multi-regional sourcing strategies (China+1), qualify backup suppliers proactively, maintain buffer inventory for high-risk materials, and map supply chain risk down to tier-2 and tier-3 suppliers. Treating diversification as ongoing operations—rather than a crisis response—is what separates resilient supply chains from vulnerable ones.
What does a resilient biopharma supply chain look like in practice?
A resilient supply chain combines diversified multi-regional sourcing, continuously monitored suppliers, and predictive digital tools for early risk detection. Critically, it also requires cross-functional alignment between procurement, quality, and regulatory teams — not just logistics infrastructure.
How is AI changing biopharma supply chain management?
AI enables supply chain teams to anticipate disruptions rather than respond to them — integrating data across suppliers, logistics, and demand patterns to inform decisions before problems escalate. In reported case studies, early adopters have seen 25% forecast error reduction and 40% lower excess inventory.


