
This guide explains how clinical trial supply chain management works, what challenges teams face in 2025, and which software platforms are leading the field—giving sponsors, research organizations, and procurement teams a practical reference.
TLDR
- CTSM spans the full lifecycle of investigational products—from forecasting and sourcing through packaging, distribution, and regulatory compliance
- Key failure points: enrollment unpredictability, cold chain breakdowns, import/export complexity, and a 50% supply waste rate across the industry
- Leading CTSM platforms now combine real-time inventory tracking, AI-driven forecasting, RTSM, and GxP compliance in one integrated tool
- For cross-border or specialty drug logistics, your distribution partner matters as much as your software platform
What Is Clinical Trial Supply Chain Management?
Clinical trial supply chain management is the end-to-end process of planning, procuring, manufacturing, packaging, labeling, distributing, and disposing of investigational medicinal products (IMPs) to ensure the right supply reaches the right site at the right time, in the right condition.
Scope Across Trial Phases
The complexity scales dramatically across phases. Phase I trials average 2 to 3 sites with small batches and narrow timelines, while Phase III trials average 24 to 58 sites spanning multiple countries, each with distinct regulatory requirements and customs procedures. Each country adds its own layer: blinded and randomized kits, multi-language labeling, and site-specific logistics coordination.
Clinical vs. Commercial Supply Chains
The practical implication: commercial supply chain playbooks — built around stable demand and long shelf lives — break down when applied to a 2-year trial where a single protocol amendment can render an entire labeling batch obsolete.
Key Components of Clinical Trial Supply Chain Management
Demand Forecasting and Inventory Planning
Demand forecasting in clinical trials is driven by enrollment rate projections, treatment duration, dropout assumptions, and protocol amendments. Inaccurate forecasting leads either to drug waste (overstock) or dangerous stockouts that halt dosing.
The Financial Toll:The median waste level for IMP kits is 50%, primarily due to poor forecasting and planning. Many investigational drugs, particularly biologics and cell therapies, have short shelf lives, making supply planning a precise calibration between excess and shortage. Insufficient inventory leads to trial delays or inability to enroll new patients, with direct costs of approximately $40,000 per day for Phase II or III trials. A single day of delay is worth approximately $800,000 in unrealized prescription drug sales.
Dynamic Forecasting Models:Traditional forecasting tools process inputs like anticipated recruitment through an algorithm to produce an average forecast, but fail to accurately factor in uncertainty. Risk-based optimization tools use Monte-Carlo simulations to model uncertain parameters—recruitment speed, patient dropout, titration probabilities—and simulate possible trial outcomes. These tools predict minimum and maximum demand, allowing teams to proceed with the lowest drug overage necessary to guarantee zero stockout risk. Implementations of these models have demonstrated 20-50% overall cost savings and 20-60% drug waste reduction.

Clinical Logistics, Distribution, and Cold Chain
Accurate demand forecasts only matter if product reaches the right site at the right time. The physical supply chain spans multiple handoffs, each with its own compliance and timing requirements:
- Manufacturing site to central depot or 3PL hub
- Central depot to investigational sites (or directly to patients in decentralized trials)
- Packaging and blinded labeling at each distribution stage
- Country-specific customs clearance and import documentation
- Last-mile delivery to site pharmacies or home healthcare settings
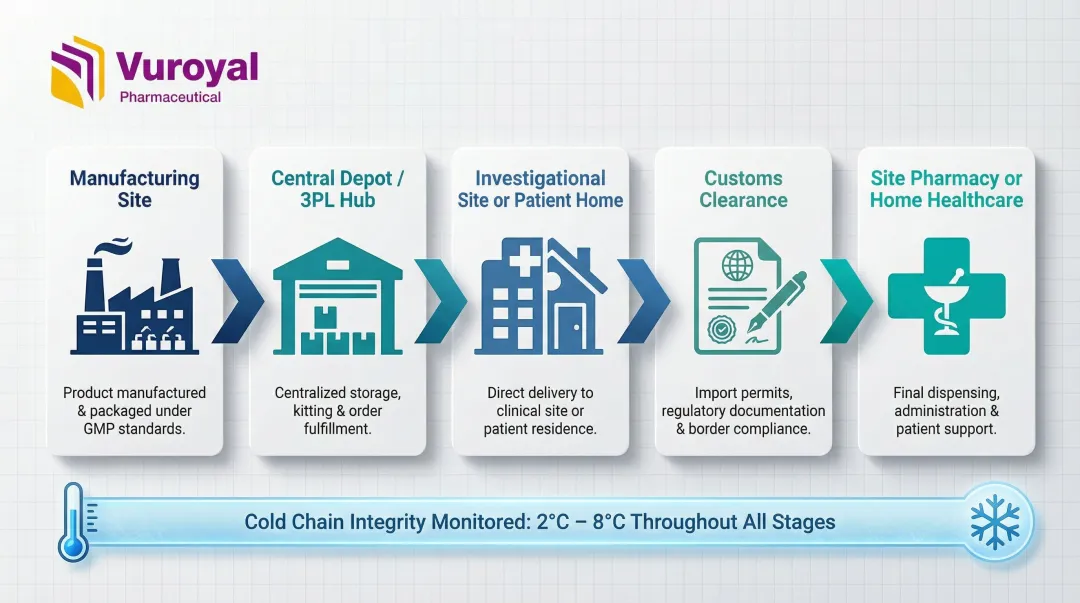
Cold Chain Criticality:Cold chain medicines grew at 13% annually between 2017 and 2022, compared to 6% for the broader pharmaceutical market over the same period. Biologics, mRNA therapeutics, and cell and gene therapies require continuous temperature monitoring from fill-finish through final delivery. A single excursion can render an entire shipment unusable — a costly outcome when investigational material is irreplaceable or in limited supply.


