
Introduction
Picture this: You're managing a Phase III trial across 80 sites in 15 countries. Every day, site coordinators enroll new participants, each requiring immediate randomization. Your investigational product (IP) inventory must be tracked in real time—some sites are overstocked while others risk running out. Blinding must remain intact despite constant dispensing activity. Temperature-sensitive biologics need cold chain monitoring. Protocol amendments arrive mid-study, requiring rapid system updates without halting enrollment.
Without a centralized technology system, this operational complexity becomes unmanageable — enrollment delays mount, IP waste accumulates, protocol deviations slip through, and trial integrity suffers. Randomization and Trial Supply Management (RTSM) systems exist to solve exactly this problem, providing the operational infrastructure that keeps multi-site trials running without breaking down.
RTSM automates patient assignment and drug logistics in a single platform, keeping trials on schedule while protecting data integrity and regulatory compliance. This guide covers RTSM's core components, terminology, key benefits, and what to look for when selecting a system.
TLDR
- RTSM automates patient randomization and investigational product logistics in a single integrated platform
- It eliminates selection bias while reducing IP waste by 20–50% through dynamic supply forecasting
- RTSM encompasses IVRS (phone-based), IWRS (web-based), and IRT — three related terms often used interchangeably
- Key benefits include faster trial execution, improved data integrity, and substantial cost savings
What is RTSM in Clinical Trials?
Randomization and Trial Supply Management (RTSM) is an integrated digital platform that automates three critical trial functions: patient enrollment, randomization to treatment arms, and investigational product supply management across all trial sites.
The acronym captures two core modules working in tandem:
- Randomization module - Assigns participants to treatment or control groups using predefined algorithms, eliminating selection bias
- Supply management module - Tracks, allocates, and resupplies investigational products throughout the trial lifecycle
Managing both functions in one system matters because randomization directly drives supply allocation. When a participant is randomized, the system must immediately assign the correct treatment kit while preserving blinding—a handoff that separate systems cannot reliably coordinate.
How RTSM Works in Practice
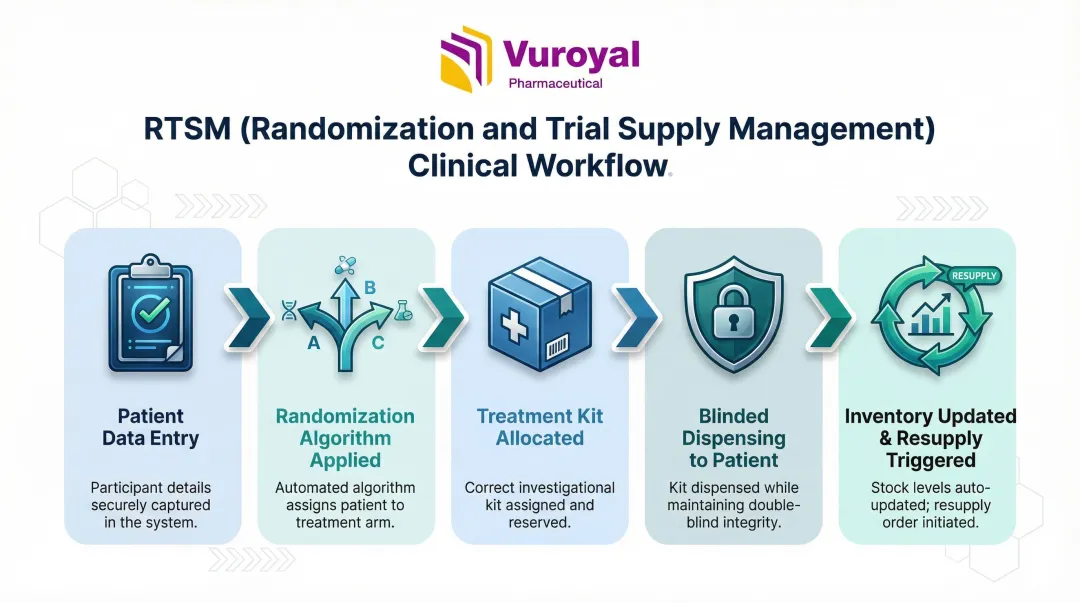
Here's a typical workflow:
- A site coordinator logs into the RTSM platform and enters a patient's eligibility data and visit information
- The system applies the protocol's randomization algorithm (simple, block, stratified, or adaptive) and assigns the participant to a treatment arm
- RTSM allocates a specific treatment kit number from available site inventory
- The coordinator dispenses the kit to the participant—without ever seeing the actual treatment assignment, preserving the blind
- The system updates inventory in real time, triggering automatic resupply orders when stock falls below predetermined thresholds
When RTSM is Deployed
RTSM is standard practice for Phase II through Phase IV trials, particularly:
- Multi-site studies requiring centralized randomization
- Blinded trials where treatment allocation must remain masked
- Complex adaptive designs with dynamic allocation probabilities
- Trials with sophisticated stratification requirements
Even smaller trials benefit from supply management automation. Preventing costly overstocking and reducing investigational product waste are pressures every trial faces, regardless of size.
Regulatory and Compliance Context
RTSM systems must align with Good Clinical Practice (GCP) guidelines and regulatory expectations from agencies including the FDA and EMA. Because RTSM directly controls unblinding, audit trails, and chain-of-custody documentation for investigational products, these systems are classified as critical computerized systems requiring strict validation.
According to the EMA's 2023 Guideline on Computerised Systems, RTSM platforms must enforce ALCOA++ principles (Attributable, Legible, Contemporaneous, Original, Accurate, Complete, Consistent, Enduring, Available, Traceable) and provide inspectors with direct read-only access to audit trails. The FDA's 21 CFR Part 11 similarly requires time-stamped, immutable audit trails where users cannot modify their own activity logs.
The Four Types of Randomization in Clinical Trials
Randomization is the gold standard for evaluating treatment efficacy because it removes selection bias, balances known and unknown confounders across groups, and makes trial results scientifically defensible. Properly executed randomization ensures each participant has an equal, pre-specified probability of assignment to any treatment arm, creating statistically valid comparisons for regulatory submissions.
The four primary randomization methods used in modern trials each suit different study designs and operational constraints. The table below provides a quick orientation before the detailed breakdown.
| Method | Best Use Case | Key Limitation |
|---|---|---|
| Simple | Large trials (n ≥ 200) | Group imbalance risk in small trials |
| Block | Multi-site or interim analysis trials | Predictable sequences if block size is guessed |
| Stratified | Trials with strong prognostic covariates | Complexity increases with number of strata |
| Adaptive | Oncology / platform studies | Implementation complexity; regulatory scrutiny |

Simple Randomization
The most straightforward method—essentially a coin flip for each participant. Each individual is assigned to a treatment group with equal probability, independent of all other assignments.
Limitation: In small trials (n < 200), simple randomization can produce group imbalances by chance, potentially undermining statistical power and baseline comparability.
Best for: Large trials where the law of large numbers ensures balance over time.
Block Randomization
Divides participants into fixed-size blocks to ensure balanced group sizes at regular intervals. For example, in a two-arm trial with block size 4, each block contains 2 participants assigned to treatment and 2 to control, but the order within each block is randomized.
Advantage: Guarantees balance even if enrollment stops unexpectedly, making it well-suited for multi-site trials where enrollment rates vary unpredictably by site.
Best for: Trials requiring interim analyses or where site-level balance is important.
Stratified Randomization
Separates participants into subgroups (strata) based on key prognostic variables—such as age, disease severity, or biomarker status—before applying randomization within each stratum. This ensures baseline balance across clinically important factors that could confound treatment effects.
A 2019 systematic review found that 69% of top-tier trials published in NEJM, JAMA, BMJ, and Lancet used stratified block randomization, making it the dominant method in modern Phase II/III studies.
It works best when specific covariates—age, gender, disease stage—are known to strongly influence outcomes.
Adaptive Randomization
A dynamic method where assignment probabilities are updated as trial data accumulates. For example, response-adaptive designs increase the probability of assigning participants to better-performing arms based on interim efficacy data.
Adoption trend: Adaptive randomization is increasingly employed in complex oncology trials and platform studies, though its use declined to just 7% in trials initiated after 2010 due to implementation complexity and regulatory scrutiny.
Critical requirement: RTSM systems are essential to execute adaptive randomization correctly, as manual implementation creates unacceptable risks of errors and unblinding.
How Trial Supply Management Works in RTSM
Trial supply management is the end-to-end tracking of investigational products from manufacturing through packaging, labeling, site distribution, dispensing, returns, and destruction. RTSM automates this entire lifecycle to ensure the right product reaches the right patient at the right time—while maintaining blinding integrity and minimizing waste.
Dynamic Inventory Forecasting Reduces Waste
Without dynamic supply management, clinical trials face 20-50% IP waste rates due to static buffer stocks, poor forecasting, and product expiration at sites. RTSM uses real-time inventory data across all trial sites to trigger automatic resupply orders, prevent stockouts, and avoid overstocking.
A Phase III case study demonstrated that RTSM-based dynamic tracking reduced drug usage by 29.5% compared to traditional pack-based planning, saving approximately €60,000 net after software costs. These savings stem from algorithm-driven forecasting that adjusts supply levels based on actual enrollment rates, dropout patterns, and visit schedules—not static assumptions.
Preserving Blinding Through Automated Kit Allocation
RTSM must simultaneously allocate treatment kits to participants while keeping the treatment assignment masked from site staff. This is achieved through:
- Scrambled kit numbering systems - Kit numbers are randomized rather than sequential, preventing site coordinators from deducing treatment patterns
- Automated dispensing logic - The system assigns kits based on randomization results without displaying treatment arm information
- Role-based access controls - Only designated unblinded personnel (typically at the sponsor or central pharmacy) can view treatment assignments

This automated approach eliminates the risk of accidental unblinding that occurs with manual supply management, where physical kit organization or documentation can inadvertently reveal treatment allocation.
Cold Chain Management for Temperature-Sensitive Products
For temperature-sensitive investigational products—including biologics, vaccines, and adjuvants—supply chain integrity depends on validated cold chain capabilities. Over 80% of biologic drugs require strict temperature-controlled conditions (refrigerated or frozen), making cold chain logistics a critical component of modern RTSM.
Advanced RTSM platforms integrate with temperature monitoring systems to automatically quarantine compromised kits based on temperature excursion data, preventing dispensing of potentially degraded product.
Sponsors managing cold chain supply for clinical trials can work with FDA-registered distributors such as VuRoyal Pharmaceutical, which provides cold chain transportation for Reference Listed Drugs and vaccine adjuvants (including QS-21 and Matrix-M™) across multi-site studies.
RTSM vs. IRT, IWRS, and IVRS: Clearing Up the Terminology
RTSM is the overarching term describing the function (randomization + supply management), while IRT, IWRS, and IVRS describe the technology delivery mechanisms used to execute those functions. All three technologies can perform RTSM functions—the difference lies in how users interact with the system.
| Term | What it Stands For | How it Works | Current Usage Status |
|---|---|---|---|
| IVRS | Interactive Voice Response System | Phone-based system where site coordinators call a toll-free number and use touch-tone keypad to randomize patients and receive kit assignments | Legacy technology from the 1990s; largely replaced by web-based systems but still used in regions with limited internet access |
| IWRS | Interactive Web Response System | Web-based platform accessed through secure browser login; site coordinators enter data via online forms and receive immediate randomization results | Modern standard; dominant technology since the early 2000s due to superior user experience and data capture capabilities |
| IRT | Interactive Response Technology | Umbrella term covering all access methods (voice, web, mobile app, SMS) used to deliver randomization and supply management functions | Current industry-standard terminology; used interchangeably with RTSM by most vendors |
| RTSM | Randomization and Trial Supply Management | Functional term emphasizing the dual capabilities (randomization + supply management) regardless of technology interface | Modern functional term; emphasizes what the system does rather than how users access it |

What is the Key Difference Between IWRS and RTSM?
IWRS is a type of technology platform used to execute RTSM processes, meaning IWRS is a subset or tool within the broader RTSM framework. RTSM defines what needs to be done—randomize patients, manage supply—while IWRS defines how users access the system to do it, specifically via web browser.
Key Benefits of RTSM for Clinical Trial Sponsors and Research Organizations
Enhanced Data Integrity and Regulatory Compliance
RTSM automates randomization and maintains tamper-proof audit trails, reducing the risk of accidental unblinding and human error in patient assignment that could compromise study validity or jeopardize regulatory approval.
RTSM systems must comply with FDA 21 CFR Part 11 and EMA ALCOA++ requirements, generating complete, time-stamped audit trails that document every user action—from patient randomization to kit dispensing to emergency unblinding events. This documentation gives sponsors defensible evidence of trial integrity during regulatory inspections.
Operational Efficiency and Faster Execution
RTSM centralizes enrollment, randomization, and supply workflows into a single system, reducing manual data entry, inter-system reconciliation errors, and site coordinator workload. This integration enables faster site activation and accelerates overall trial execution.
Integration capability is a deciding factor for most buyers: 61% of sponsors and CROs rank EDC, ePRO, and CTMS connectivity as a top-5 selection criterion. Seamless data flow between RTSM and Electronic Data Capture (EDC) systems eliminates duplicate entry and enables real-time monitoring of enrollment progress, protocol deviations, and supply status.
Substantial Cost and Waste Reduction
Automated resupply algorithms and drug pooling features (where inventory can be shared across sites without manual vendor intervention) reduce over-supply and expiration waste. According to industry analysis, optimized RTSM forecasting reduces supply waste by 15–30%, translating to hundreds of thousands of dollars in savings for large Phase III trials.

For biologics requiring cold chain management, the impact is even greater. Temperature-sensitive products can cost tens of thousands of dollars per batch — preventing even a single waste event justifies the investment.
What to Look for in a Modern RTSM System
Not all RTSM systems are built for the demands of modern, multi-site trials. These three criteria help separate systems that simply meet minimum requirements from those that actively support trial efficiency.
Integration Capability with Trial Tech Stack
A strong RTSM system should connect with your trial's EDC, eCOA (electronic Clinical Outcome Assessment), and CTMS (Clinical Trial Management System) without requiring manual data reconciliation. Siloed systems create data reconciliation burdens, increase the risk of transcription errors, and slow database lock.
When evaluating vendors, ask:
- Does the vendor offer native integration with your existing EDC platform?
- Do they provide robust APIs for custom integration?
- Do they offer a unified platform where RTSM is built into the EDC, or a standalone system requiring separate integration work?
Speed and Configurability for Protocol Amendments
76% of protocols experience at least one substantial amendment, taking an average of 260 days to fully implement. Given this reality, prioritize systems that support agile setup and rapid mid-study updates.
Look for systems that allow protocol changes without re-validating the entire system from scratch. Specifically, the ability to:
- Modify dosing schedules mid-study
- Add or adjust stratification factors
- Update visit windows without weeks of downtime
Natural language processing-based configuration tools and rapid build frameworks are worth asking vendors about directly.
Vendor Track Record and 24/7 Support
Because RTSM directly controls patient safety and drug dispensing, any system downtime or configuration error has immediate trial consequences. Evaluate vendor track record, helpdesk availability, and the ability to handle multi-regional, multi-language deployments for global trials.
Before committing to a vendor, get specific answers on:
- Average system uptime percentage
- 24/7 technical support coverage across all time zones
- References from sponsors running similar trial designs
- Process for emergency unblinding and system recovery
Frequently Asked Questions
What is randomization and trial supply management?
RTSM is a clinical trial technology system with two core functions: automated patient randomization and trial supply management. Randomization assigns participants to treatment or control groups; supply management tracks and allocates investigational products. Together, they keep trials efficient, unbiased, and low on IP waste.
What is the purpose of randomization in drug trials?
Randomization eliminates selection bias by giving each participant an equal probability of assignment to any treatment arm. This balances known and unknown confounders across groups, making outcomes statistically valid and credible for regulatory submissions.
What are the 4 types of randomization?
The four main types are:
- Simple randomization — coin-flip method, no constraints
- Block randomization — ensures balance at regular intervals
- Stratified randomization — balances key prognostic factors across groups
- Adaptive randomization — adjusts assignment probabilities based on accumulating data
The right choice depends on trial size, design complexity, and whether subgroup balance is critical.
What is the difference between IWRS and RTSM?
RTSM describes the overall function (randomization + supply management), while IWRS is a web-based technology platform used to execute those functions. IWRS is one type of tool within the broader RTSM category—not a different concept. RTSM defines the capability; IWRS is the system that delivers it.
How does RTSM improve data integrity in clinical trials?
RTSM improves data integrity by automating patient assignment to eliminate manual errors and preserving blinding through coded kit dispensing. It also generates complete, tamper-proof audit trails that document every system action with timestamps and user attribution, meeting GCP and FDA requirements.
When is RTSM required in a clinical trial?
While not always mandated by regulation, RTSM is considered best practice for any randomized, controlled trial—especially Phase II–IV studies, multi-site trials, blinded studies, or trials requiring complex supply chain management across geographies. Even smaller trials benefit from automated supply forecasting to reduce IP waste.


