
Introduction
Clinical trial packaging operates under fundamentally different pressures than commercial pharmaceutical packaging. Commercial packaging optimizes for shelf appeal and cost efficiency. Clinical supply packaging must protect product integrity, maintain blinding, satisfy multi-country regulatory requirements, and support complex protocols—while absorbing protocol amendments and supply chain volatility as they happen.
The stakes are disproportionately high. A French simulation study found a 12% error rate in investigational drug container labels, while cold chain logistics report 2-5% shipment failures from temperature excursions. These failures don't just waste materials—they invalidate data, trigger regulatory holds, and compromise patient safety.
54% of Phase III trials fail in clinical development, with 57% of those failures linked to inadequate efficacy—often confounded by poor medication adherence tied to packaging failures. That makes packaging a critical study design element, not a procurement afterthought.
This article covers the types of packaging used in clinical trials, FDA and international regulatory requirements, key design considerations for kits and cold chain materials, emerging trends in smart packaging and just-in-time models, and best practices for sourcing and supply chain resilience.
TLDR:
- Clinical supply packaging must protect integrity, maintain blinding, and satisfy multi-country regulations simultaneously
- Primary packaging is shifting from glass to polymers like COP to prevent delamination in biologics
- FDA mandates specific labeling elements (IND number, storage conditions, investigational use statement) under 21 CFR Parts 211 and 312
- Cold chain failures cost biopharma approximately $35 billion annually, making validated thermal packaging non-negotiable
- Just-in-time labeling and demand-led supply models cut product destruction and shipments by up to 40%
What Are Clinical Trial Supplies and Why Does Packaging Matter?
Clinical supplies encompass all investigational drug products, comparator drugs, placebos, and ancillary materials required to conduct a clinical trial. Unlike commercial pharmaceuticals, these materials must support research protocols that demand dose anonymity, multi-country distribution, and the flexibility to accommodate protocol amendments mid-study.
Packaging failures carry consequences far beyond wasted materials. The biopharma industry loses approximately $35 billion annually due to temperature-controlled logistics failures. A labeling error can introduce bias that invalidates months of data collection.
Temperature excursions create a separate category of risk. A degraded product can compromise patient safety and trigger FDA clinical holds — the agency will place an IND on clinical hold if a product cannot remain chemically stable throughout the testing program or if it presents an impurity profile indicative of a health hazard.
Packaging requirements vary significantly by trial phase, drug type, and geography:
- Phase I vs. Phase III: Phase I investigational drugs are exempt from full commercial cGMP regulations in 21 CFR Part 211, provided they follow specific FDA Phase 1 CGMP guidance focusing on safety and orderly manufacturing. Phase III studies, by contrast, require large quantities with formulations close to the intended commercial product — consistency and scalability become non-negotiable.
- Small molecule vs. biologic: Traditional small molecules tolerate standard glass vials. High-pH biologics are vulnerable to glass delamination, driving demand for alternative materials like cyclic olefin polymers (COP).
- Single-country vs. multinational: Domestic trials require English-language labeling and FDA compliance. Multinational trials must simultaneously satisfy FDA, EMA (EU CTR/Annex 13), ICH Q10, and country-specific regulations — requirements that need to be designed in from the start, not retrofitted mid-study.

Getting packaging strategy right at the design stage is what separates trials that run on schedule from those that face costly amendments or regulatory delays.
Types of Packaging Used for Clinical Supplies
Primary Packaging
Primary packaging refers to the container in direct contact with the drug product—vials, blister packs, prefilled syringes, autoinjector pens, and ampoules. The choice of primary container is driven by dosage form, drug stability, and route of administration.
Clinical trial pipelines are increasingly focused on parenteral and biologic delivery systems. Over half of all clinical trials in Phases I through III are currently investigating parenteral formulations, driving demand for prefilled syringes and autoinjectors. The global prefilled syringes market is projected to grow at a 9.7% CAGR to reach $18.12 billion by 2033, while autoinjectors are growing at 13.8% CAGR.
Greater reliance on biologics sharpens the stakes for material compatibility. High-pH biologics are vulnerable to glass delamination, which leaches glass flakes into drug products and triggers recalls. Polymer alternatives address this directly:
- Cyclic olefin polymer (COP) syringes — eliminate silicone-oil interactions that cause protein aggregation, with high break resistance and a superior moisture barrier
- Polypropylene — a cost-accessible alternative to borosilicate glass for less reactive formulations
Sponsors must validate material selection against the specific drug formulation through rigorous extractables and leachables (E&L) testing per USP <1664> and ICH Q3E guidelines.
Secondary Packaging
Secondary packaging — cartons, blister overwraps, and clinical kits — forms the outer layer that groups and protects primary containers. It handles breakage protection during shipping, temperature control, and regulatory labeling, often serving all three functions simultaneously.
For decentralized and direct-to-patient (DTP) studies, secondary packaging also serves a patient-communication function. Clear instructions, inserts, and dividers improve usability when patients self-administer investigational products at home without direct clinical supervision. The kit must anticipate questions, provide visual cues for sequencing, and accommodate patients with limited dexterity.
Tertiary and Shipping Packaging
Tertiary packaging—shipping containers, shippers, and temperature-controlled packaging systems—is the outermost layer responsible for protecting shipments during distribution. For cold-chain materials, the tertiary system is not simply a box; it's a validated thermal performance solution.
Validated thermal packaging must maintain specific temperature ranges (2°C–8°C refrigerated, -20°C or -80°C frozen) for the expected transit duration. Testing standards like ISTA 7D/7E and ASTM D7386 provide frameworks for evaluating insulated shipping containers under real-world transit conditions. These validations document that the packaging system can maintain temperature integrity even when exposed to external temperature extremes during international shipment.
FDA and Regulatory Requirements for Clinical Supply Packaging
The FDA requires clinical trial packaging to comply with 21 CFR Parts 211 and 312, which govern current Good Manufacturing Practice (cGMP) standards for investigational new drugs (INDs). These regulations cover container closure integrity, labeling content, and packaging controls.
Required Labeling Elements:
Under 21 CFR 312.6, the immediate package of an investigational new drug must bear the exact statement: "Caution: New Drug—Limited by Federal (or United States) law to investigational use." Additional required elements include:
- IND number
- Protocol number
- Dosage instructions
- Storage conditions
- Sponsor name and address
- Statement identifying the product as investigational
All labeling must appear in English; multi-country trials also require local-language labels per each country's health authority.
Serialization and Blinding Protection:
For randomized controlled trials, unique identifiers (serial numbers) must be used to protect blinding. The unblinding key must be held by the labeler separately from the serialized label assignment to prevent inadvertent unblinding. While commercial drugs require strict serialization under the Drug Supply Chain Security Act (DSCSA), investigational drugs are exempt under section 581(24) of the FD&C Act to protect dose anonymity in blinded trials.
Multinational Regulatory Complexity:
For multinational trials, sponsors must simultaneously satisfy FDA requirements alongside EMA (EU CTR/Annex 13 of EU GMP), ICH Q10, and country-specific regulations.
The EU Delegated Regulation 2022/2239 amended Annex VI to allow sponsors to omit the expiry date on immediate packaging when it remains with the fully labeled outer packaging or takes the form of small units like blisters. This reduces the costly, product-degrading cycle of frequent relabeling to update expiry dates.
EU GMP Annex 13 also requires intensified precautions against mislabeling for blinded products, noting that IMP packaging and labeling are more complex and error-prone than marketed products. Key regulatory areas sponsors must plan for from the outset include:
- Simultaneous FDA and EMA compliance for blinded trial packaging
- Country-specific labeling language requirements
- Expiry date management across jurisdictions
- Mislabeling controls unique to investigational materials

Managing these overlapping requirements is one area where sponsors frequently encounter delays. VuRoyal offers free regulatory guidance to help clinical trial sponsors work through multi-jurisdictional documentation, imported drug approval registration, and compliance verification before packaging decisions are finalized.
Key Packaging Considerations for Clinical Trial Materials
Kit Design and Patient-Centric Packaging
Clinical kit design must be built around how investigators and patients will actually use the materials. Critical considerations include:
- Sequence of access: Items must be arranged in the order they'll be used
- Ancillary supplies: Syringes, needles, wipes, sharps containers must be included and properly organized
- Physical dimensions: Kits must fit within standard shippers to control shipping costs
Poor kit planning can exponentially increase costs through inefficient use of shipper volume, repeated shipments, or site complaints that trigger repackaging.
Decentralized clinical trials (DCTs) and direct-to-patient (DTP) shipping have reshaped kit design. Kits must now incorporate drug product, administration supplies, patient instructions, and return packaging in a single unit. This increases kit complexity compared to site-based trials, where clinical staff provide guidance and handle ancillary materials separately.
Child-resistant and tamper-evident features are required under the Poison Prevention Packaging Act (PPPA) codified in 16 CFR 1700. These features must be balanced with accessibility for patients who may have limited dexterity, particularly elderly participants or those with arthritis.
Cold Chain and Temperature Control
Biologic drugs—monoclonal antibodies, vaccines, gene therapies, cell therapies—typically require cold chain packaging validated for specific temperature ranges. The tertiary packaging system must be validated to maintain these conditions for the expected transit duration, accounting for worst-case scenarios like shipping delays or extreme ambient temperatures.
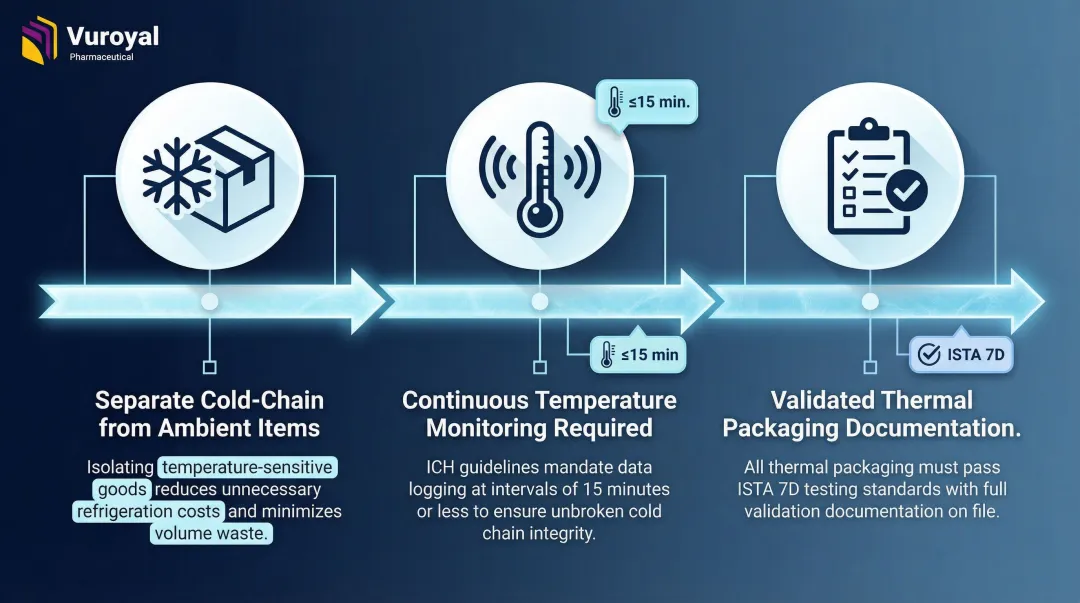
Key cold chain principles:
- Separate cold-chain drug product from ambient ancillary items: Shipping ancillaries (syringes, wipes, instructions) inside a cold-chain shipper inflates cost and cuts available volume. For site-based trials, a separate ambient ancillary kit is often the better solution.
- Temperature monitoring is required: Data loggers, time-temperature indicators, and IoT sensors document excursion-free delivery. ICH guidelines mandate continuous monitoring at ≤15-minute intervals for refrigerated and frozen shipments.
- Validation must be documented: Thermal packaging must undergo ISTA 7D or equivalent testing to demonstrate it maintains target temperature ranges under real transit conditions.
VuRoyal supports clinical trial sponsors with cold chain supply chain services for temperature-sensitive pharmaceutical materials, including reference drugs and vaccine adjuvants requiring controlled-temperature transit.
Labeling Best Practices
Clinical trial labels must maintain batch-to-batch consistency across all print runs and facilities. Even minor inconsistencies in secondary packaging labels risk introducing bias that could affect blinded study outcomes.
Best practices include:
- Automated vision inspection systems: These have replaced manual inspection in many clinical packaging facilities, improving both consistency and throughput while reducing human error
- Centralized label artwork management: A single source of truth for label artwork prevents version control errors
- 100% label verification: Per 21 CFR Part 211 Subpart G, labels must undergo 100% examination for correct labeling or automated differentiation to prevent mix-ups
Blinding and Comparator Drug Packaging
For randomized blinded trials, packaging must visually conceal whether a participant is receiving the investigational product or the comparator/placebo. This typically requires:
- Over-encapsulation to mask differences in tablet appearance
- Custom blister card design with identical appearance across treatment arms
- Identical secondary packaging for all treatment arms
Sourcing and repackaging comparator drugs adds complexity. The original commercial packaging must be replaced with study-specific packaging that meets IND requirements without altering product stability. This requires stability studies to demonstrate that repackaging doesn't compromise drug integrity.
Emerging Trends Shaping Clinical Supply Packaging
Smart Packaging Technologies
NFC (near-field communication) tags, RFID labels, barcodes, and connected devices support patient adherence monitoring, real-time temperature tracking, and supply chain authentication. Smart packaging achieves 97% accuracy in tracking adherence, compared to 60% for pill counting and just 27% for self-reporting. NFC tags embedded in packaging can wirelessly communicate with a patient's smartphone to verify correct drug intake and provide dosing reminders.
Late-Stage Customization (LSC) and Just-in-Time (JIT) Packaging
Traditional mass packaging results in massive overage, waste, and costly relabeling efforts. The JIT model holds drug product unpackaged or in bulk until demand signals from clinical sites trigger final packaging and labeling.
Demonstrated benefits (Catalent demand-led supply model case study):
- Reduced product destruction by 40%
- Cut total shipments by 40%
- Accelerated first-patient dosing to under three weeks
- Same-day delivery (24–36 hours) from demand signal to shipment

These efficiency gains are further supported by Digital Display Labels (DDLs), which use e-paper and NFC technology to eliminate manual relabeling entirely — updating expiry dates via automated system controls in days rather than weeks.
Selective Automation in Kitting
Clinical kitting remains largely manual due to volume variability and kit diversity. Automation is being adopted selectively where it delivers the clearest gains:
- High-volume, standardized kits: Automated lines handle consistent configurations at scale in GMP cleanrooms
- Multi-product lines: Modular automation supports packaging and labeling of multiple drugs without cross-contamination
- JIT integration: Automated kitting lines connect directly to demand-signal workflows, reducing lag between order and shipment
Sourcing and Supply Chain Best Practices
Secure Supply Agreements Early
The availability of packaging materials—primary containers, ancillary items, temperature monitors, and custom inserts—directly affects trial timelines. The COVID-19 pandemic highlighted this vulnerability: lead times for glass vials grew from 3-6 months to 18 months as manufacturers pivoted to vaccine production.
Sponsors should:
- Secure supply agreements early in trial planning
- Identify secondary sources for critical materials to avoid single-source dependency
- Account for extended lead times when planning trial timelines
Work with Integrated, Single-Source Partners
Using multiple vendors across the supply chain creates handoff risks and communication gaps. An integrated partner that can handle clinical supply procurement, cold chain logistics, customs clearance, and regulatory compliance reduces these risks.
VuRoyal Pharmaceutical is an FDA-registered, Massachusetts-licensed wholesaler with customs declaration credentials and drug business quality management certification — covering international procurement, import approval registration, customs clearance, and cold chain transportation under a single point of accountability.
Build Supply Chain Resilience for International Trials
Multinational trials introduce additional complexity:
- Customs clearance timelines: Account for country-specific processing times and potential delays
- Import regulations: Each country has unique requirements for investigational medicinal products
- Country-specific documentation: Certificates of origin, import permits, and Qualified Person (QP) certifications vary by jurisdiction
Sponsors should map these requirements during protocol development, not after enrollment begins. A partner with established country-specific regulatory knowledge reduces the risk of documentation gaps that can delay site activation by weeks.
Frequently Asked Questions
What is a clinical supply?
Clinical supplies are all investigational drug products, comparator drugs, placebos, and ancillary materials required to conduct a clinical trial. They must be manufactured, packaged, and labeled under cGMP standards per FDA regulations.
What are the FDA requirements for clinical supply packaging?
FDA requires compliance with 21 CFR Parts 211 and 312. Required label elements include the IND number, storage conditions, and the "Caution: New Drug—Limited by Federal Law to Investigational Use" statement. Serialization requirements also apply to protect blinding in randomized trials.
What types of packaging are used for clinical supplies?
Clinical supplies use three packaging levels:
- Primary (vials, syringes, blister packs) — contacts the drug directly and protects its physical and chemical integrity
- Secondary (cartons, clinical kits) — groups primary containers and carries patient and regulatory labeling
- Tertiary/shipping (temperature-controlled shippers) — protects shipments during distribution
How are clinical supply kits assembled for randomized trials?
Clinical supply kits are assembled according to randomization schedules generated by IRT/IWRS systems. Each kit is assigned a unique kit number that maintains blinding — site staff dispense by kit number without knowing treatment assignment. Proper kit design must account for dosing regimens, visit schedules, and return materials.
How does direct-to-patient shipping affect clinical supply packaging?
DTP shipping increases kit complexity. Each patient-facing unit must combine:
- Drug product and administration supplies
- Clear patient instructions for use
- Return packaging for unused or used materials
Design emphasis shifts toward patient-friendly labeling and tamper-evident closures, since site pharmacists are no longer present to guide patients.
Why is cold chain packaging critical for biologic clinical trial materials?
Biologics — including vaccines, monoclonal antibodies, and cell/gene therapies — degrade rapidly outside their required temperature ranges (typically 2–8°C refrigerated or −20°C to −80°C frozen). Validated cold chain packaging and documented temperature monitoring are essential to protect product potency and patient safety.


