
Introduction
Clinical trial sourcing—the process of acquiring investigational products, comparator drugs, lab services, trial sites, and logistics infrastructure—is one of the most complex and underplanned aspects of running a successful trial. Yet sourcing failures consistently rank among the leading drivers of trial delays and budget overruns. According to the Tufts Center for the Study of Drug Development, a single day of delay in Phase II or III trials costs approximately $40,000 in direct operational costs and $800,000 in lost prescription sales — totaling $840,000 in losses per day.
VuRoyal Pharmaceutical's founders witnessed this challenge firsthand. After decades in pharmaceutical R&D and clinical research, they saw qualified organizations repeatedly fail to secure safe, genuine, high-quality substances on time — a gap that motivated them to build VuRoyal as an FDA-registered wholesaler with supply chain capabilities covering procurement, cold-chain logistics, and regulatory clearance for reference listed drugs and vaccine adjuvants.
What follows is a practical breakdown of the sourcing decisions that most directly affect trial timelines, data integrity, and cost — so sponsors, CROs, and procurement teams can act before delays become crises.
TLDR
- Clinical trial sourcing covers investigational drugs, comparators, lab services, logistics, and regulatory compliance—each element directly affects trial outcomes
- Define your sourcing strategy during protocol development—waiting until after ethics approval routinely causes costly, avoidable delays
- Trial design determines packaging, storage, and distribution requirements, so sourcing teams must be part of early protocol discussions
- Failures in cold chain integrity, regulatory compliance, or supply chain reliability put patient safety and trial data at risk
- FDA-registered distributors reduce regulatory burden and logistical complexity, particularly when sourcing specialty or controlled substances
What Is Clinical Trial Sourcing and Why Does It Matter?
Clinical trial sourcing is the full-cycle process of identifying and securing all materials, services, and resources required to conduct a clinical trial. This includes investigational medicinal products (IMPs), comparator drugs, laboratory services, trial sites, logistics infrastructure, and data management tools.
Core sourcing categories include:
- Drug and intervention procurement (IMPs, comparators, placebos)
- Site selection and management
- Laboratory services and sample processing
- Patient recruitment and referral partnerships
- Logistics and supply chain operations
- Regulatory compliance and documentation
- Data management systems and technology platforms
Each of these categories is only as strong as the sourcing decisions behind it. Poor vendor selection, supply delays, and non-compliant materials compromise data integrity, endanger patient safety, and delay regulatory approval.
The regulatory record makes this clear. Analysis of FDA warning letters issued between 2010–2020 found that 20–25% cited documentation and data integrity failures as major deficiencies. Inadequate IMP accountability has also appeared repeatedly in the EMA's top 10 critical GCP inspection findings.

When Sourcing Planning Must Begin
Sourcing planning must start during protocol development — before IRB or ethics committee approval. Late planning is one of the most avoidable causes of trial disruption.
Packaging and labeling booklet labels for multinational trials alone requires approximately 30 weeks of lead time. Treating comparator sourcing or ancillary supply procurement as an afterthought routinely pushes trial start dates by months.
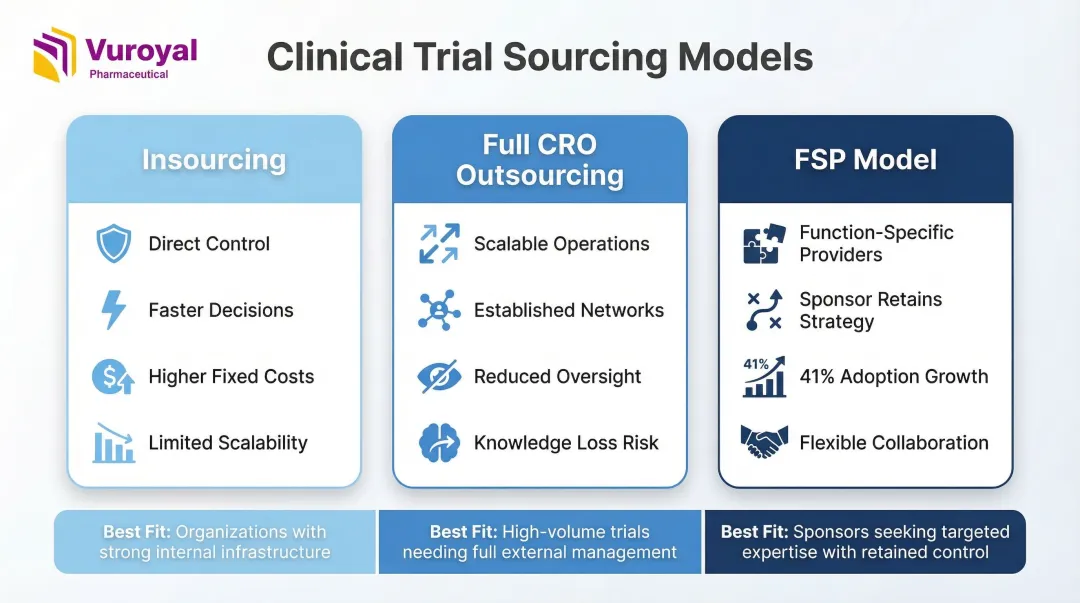
Choosing the Right Sourcing Model: Insourcing, Outsourcing, and FSP
No single sourcing model is universally optimal. The right approach depends on trial phase, company size, therapeutic area complexity, and internal capabilities.
Insourcing (In-House Clinical Operations)
Insourcing means the sponsor retains internal staff to manage key trial functions such as site monitoring, drug accountability, sample tracking, and clinical oversight.
Where insourcing excels:
- Direct control over timelines and quality
- Better protocol knowledge retention
- Faster decision-making without vendor intermediaries
- Cultural alignment with company objectives
Where it falls short:
- Requires upfront investment in infrastructure (SOPs, work instructions, staff)
- Limited scalability for rapidly expanding trials
- Higher fixed costs compared to variable CRO pricing
Outsourcing to CROs
Where insourcing hits its limits, Contract Research Organizations (CROs) pick up the slack. Full outsourcing offers scalability, access to established networks, and cost efficiency — but sponsors often lose direct oversight, institutional knowledge, and alignment when CRO staff are spread across multiple studies.
Best practice: Assign an experienced in-house study manager to govern CRO performance against Statement of Work (SOW) deliverables, KPIs, and timelines. This maintains strategic control while leveraging external infrastructure.
The FSP (Functional Service Provider) Model
According to PPD's 2023 Pulse Report, FSP outsourcing is growing faster than full-service models—41% of respondents reported increased FSP use compared to just 29% for full-service outsourcing. The FSP model engages specialized external providers for specific functions (data management, biostatistics, monitoring) while the sponsor retains strategic oversight.
Phase I and Phase II trials especially benefit from a combined insourcing/FSP approach that keeps clinical operations knowledge in-house while flexibly scaling specialized roles. This model lets sponsors maintain control over their data and systems without the rigid lock-in of a traditional full-service CRO contract.
Decision framework: Critically evaluate each trial function and assign it to the model that best balances control, expertise, cost, and risk. For most early-phase sponsors, the answer is rarely full outsourcing — it's a deliberate mix.
How Trial Design Influences Your Sourcing Approach
Trial design determines how investigational products, comparators, and concomitant medications must be sourced, packaged, stored, and distributed — which is why sourcing teams need a seat at the table during protocol design, not after it.
Blinded Trials: The Five Senses Challenge
Blinded trials (single-blind and double-blind) require products to be blinded in both packaging and documentation. Under ICH E6(R3) and EU GMP Annex 13, IMPs must be coded and labeled to prevent inappropriate unblinding.
Sourcing complexities:
- Products must match across all five human senses—sight, smell, taste, feel, and weight
- Biologics and sterile injectables require opaque IV bags or tinted syringes to mask visual differences
- Specialized packaging complicates import/export processes
- Shipping and storage conditions across treatment arms must be equalized to preserve blinding
- Interactive Response Technology (IRT) systems provide emergency unblinding procedures for medical emergencies
Open-Label Trials: Simplified Sourcing
Open-label trials have no blinding requirements, allowing for:
- Local sourcing of comparators through regional wholesalers
- Reduced repackaging needs and simpler import/export documentation
- Shorter lead times and lower costs
- Direct procurement from local pharmaceutical distributors
Add-On and Randomized Trials
These trial types introduce distinct sourcing obligations that must be addressed early in planning:
- Add-on studies require compatibility testing and safety documentation for combination treatments — adverse interaction risks between the IMP and concomitant medications must be evaluated before procurement begins
- Randomized trials demand that suppliers receive full participant counts per arm, dosage quantities, and site distribution details; without this data, accurate forecasting breaks down and both stockouts and wastage become likely
Best Practices in Clinical Trial Supply Chain and Logistics
Start Early with Comprehensive Planning
Develop a logistics plan that covers shipping routes, storage requirements, inventory control, and contingency protocols for disruptions—especially in multi-site or international trials. Supply chain planning is a critical path activity, not an administrative afterthought.
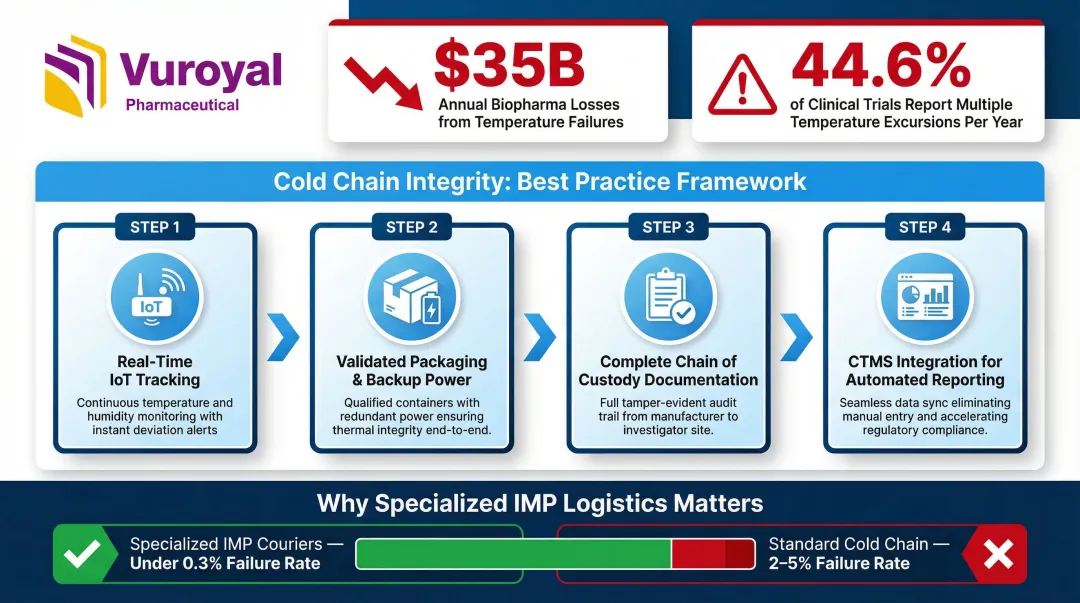
Cold Chain and Temperature-Controlled Logistics
The biopharma industry loses approximately $35 billion annually due to failures in temperature-controlled logistics. A 2019 IQVIA survey revealed that 44.6% of respondents reported multiple temperature excursions per year, with 41% exceeding four degrees.
Critical practices:
- Upgrade from passive data loggers to real-time IoT temperature tracking
- Use validated packaging with backup power storage
- Document complete chain of custody
- Integrate IoT temperature loggers with Clinical Trial Management Systems (CTMS) for automated excursion reporting and quarantine
Because investigational products often lack approved stability data, any excursion outside specifications can invalidate a trial batch, requiring expensive replacements and delaying patient care. Specialized IMP couriers achieve failure rates below 0.3%, compared to 2-5% for standard cold chain logistics.
Demand Planning and Inventory Management
Traditional clinical supply forecasting results in a median waste level of 50% for IMP kits. Accurate forecasting based on patient recruitment timelines, site activation schedules, and protocol amendments reduces stockouts and overage waste.
Just-In-Time (JIT) inventory advantages:
- Pools supplies and executes late-stage, on-demand labeling at the point of distribution
- Drastically reduces waste from early commitment to specific sites
- Shortens timelines and lowers investment in drug destruction
- Requires IRT/RTSM systems to predict expiration dates and automate buffer stock levels
Regulatory Compliance Requirements
GCP and GDP (Good Distribution Practice) guidelines govern how materials are handled, stored, and distributed. For cross-border trials, sponsors must navigate import/export regulations, customs documentation, and country-specific drug approval requirements.
Key regulatory frameworks:
- ICH E6(R3): Sponsor responsibility for IMP manufacturing, blinding, labeling, and site accountability
- EU GDP 2013/C 343/01: Temperature control, transportation validation, and prevention of falsified medicines
- FDA 21 CFR Part 312: IND requirements, import/export rules, and mandatory "Caution: New Drug" labeling
- WHO TRS 992, Annex 5: Safe storage and distribution of time- and temperature-sensitive pharmaceutical products
Navigating these frameworks across multiple jurisdictions is one of the most operationally complex aspects of international trial supply. Partnering with an FDA-registered distributor that holds active drug business licenses, quality management certifications, and customs declaration credentials—like VuRoyal Pharmaceutical—lets sponsors offload import approval registration, customs clearance, and cold chain transportation to a single qualified source rather than coordinating across multiple vendors.
Overcoming Common Sourcing Challenges
Vendor Selection and Quality Assurance
Identifying vendors who can reliably deliver high-quality, compliant materials is one of the most persistent sourcing challenges.
- Conduct thorough due diligence including regulatory certifications (FDA registration, GMP/GDP compliance)
- Review track record, references, and financial stability
- Request proposals from multiple vendors
- Define quality agreements upfront before contract finalization
- Verify Certificates of Analysis (CoA) and Batch Release Certificates to ensure comparator drugs are consistent in quality across batches
VuRoyal employs third-party drug supply chain compliance solutions to ensure continuous compliance with state and federal regulations, providing assurance through independent oversight. VuRoyal's GDP Standard Operating Protocol follows guidelines from the U.S. Pharmacopeia (USP), Title 21 CFR, PDMA, and DSCSA.
Regulatory and Cross-Border Complexity
Modern Phase III trials involve a median of 40 sites and 1,202 participants across 84 countries. This multinational footprint introduces severe customs and regulatory bottlenecks.
- Engage regulatory experts early in planning
- Maintain meticulous documentation
- Partner with distributors that hold appropriate import/export licenses and customs registration for each operating country
- Plan for EU Qualified Person (QP) release strategies—shipping IMPs into the EU without pre-planned QP release guarantees customs holds
For sponsors managing cross-border complexity, VuRoyal's regulatory support services cover:
- Imported drug approval registration and import filing
- Customs clearance and provision of required approval documents, including the 'Imported Drug Approval Document'
- Reference preparation selection and technical consultation (available as a free consultation)
This end-to-end support helps sponsors avoid the documentation gaps that most commonly cause customs holds and approval delays.
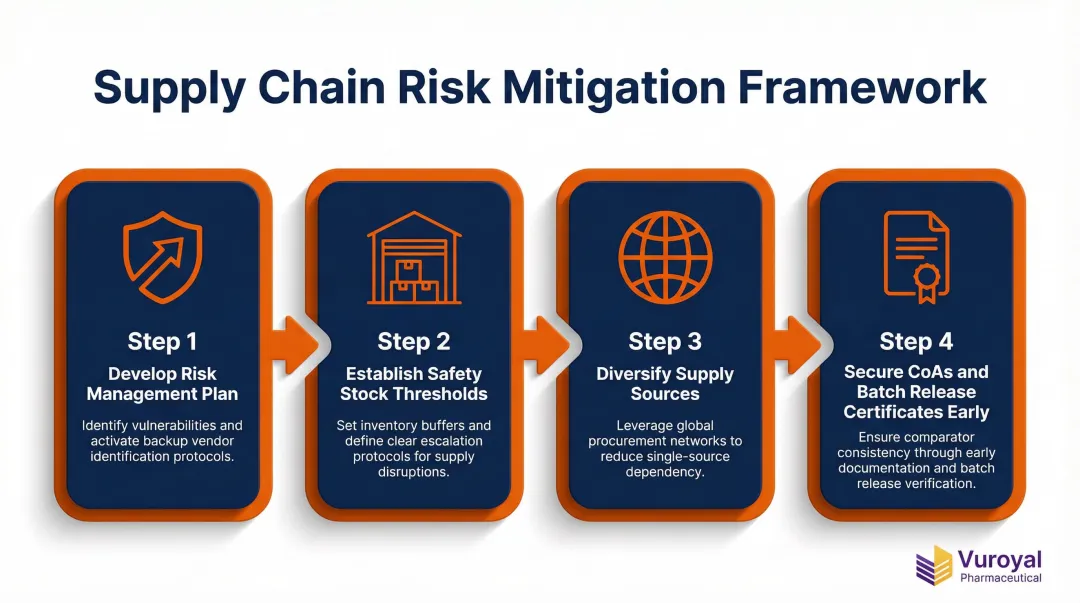
Supply Chain Disruption and Risk Mitigation
Single-source dependencies, geopolitical disruptions, and manufacturer delays can halt trial supply. Surging commercial demand for drugs like GLP-1 agonists and oncology therapies has created severe market scarcity for comparators.
- Develop a risk management plan that includes backup vendor identification
- Establish safety stock thresholds with clear escalation protocols
- Diversify supply sources and work with distributors that have established global procurement networks
- Secure CoAs and Batch Release Certificates early to verify comparator consistency
As the exclusive U.S.-China partner of Desert King International, VuRoyal provides access to premium vaccine adjuvants including QS-21 (GMP Standards), Matrix-M™, and related adjuvant technologies. Desert King holds hundreds of grams of current valid GMP-grade QS-21 inventory. Manufacturing facilities across Chile, Mexico, and Italy provide the geographic diversification that protects sponsors against single-region supply disruptions.
Frequently Asked Questions
What is the difference between insourcing and outsourcing in clinical trials?
Insourcing keeps trial functions within the sponsor's own team for greater control and knowledge retention, while outsourcing delegates these functions to external CROs for scalability. Most sponsors benefit from a hybrid approach that balances direct oversight with flexible capacity through Functional Service Provider (FSP) models.
When should clinical trial sourcing planning begin?
Sourcing planning must start during the study planning and protocol development phase, ideally before ethics approval is sought. This allows adequate time for vendor selection, regulatory compliance preparation, and logistics setup, preventing the costly delays that result from treating sourcing as an afterthought.
What types of products typically need to be sourced for a clinical trial?
Core categories include investigational medicinal products (IMPs), comparator drugs, placebos, laboratory supplies, and ancillary materials such as packaging and storage equipment. Specialty items like vaccine adjuvants or reference listed drugs require distributors with specialized regulatory credentials and cold chain capabilities.
How does trial design affect comparator sourcing?
Blinded trials require matching packaging and documentation to maintain blinding during shipping and storage, increasing complexity and cost. Open-label trials allow local sourcing at lower cost and complexity.
What regulatory standards govern clinical trial supply chains?
Clinical supply chains must comply with Good Clinical Practice (GCP) and Good Distribution Practice (GDP) guidelines, as well as country-specific import/export regulations. In the U.S., FDA requirements for pharmaceutical distributors include 21 CFR Part 312 for investigational drugs and 21 CFR Part 211 for cGMP compliance.
How can sponsors reduce supply chain risk in clinical trials?
Early planning, multi-source vendor strategies, and safety stock protocols are essential. Working with experienced distributors that have proven global logistics capabilities, regulatory compliance track records, and established procurement networks reduces exposure to single-point failures and geopolitical disruptions.


