
Introduction
For clinical trial sponsors running multi-country studies, a single sourcing misstep can compromise trial validity, patient safety, and regulatory standing across every jurisdiction involved. Comparator sourcing is the process of procuring approved, commercially available drugs to serve as reference benchmarks against an investigational product — spanning multiple countries, regulatory frameworks, and supply chains simultaneously.
It's a discipline that demands strategic planning, not just logistics coordination.
Though often treated as a downstream logistics task, comparator sourcing is a high-stakes, strategy-dependent discipline where a single day of delay costs approximately $800,000 in lost prescription sales, alongside $40,000 in daily trial operating costs. This guide explains what the process involves, how the main sourcing models work, and what factors determine the right approach for your trial.
TLDR
- Comparator drugs are approved medicines used to measure an investigational drug's safety and efficacy against the current standard of care
- Global sourcing requires navigating divergent regulatory requirements, import rules, supply availability, and documentation standards across multiple jurisdictions at once
- Four sourcing approaches exist (direct from manufacturer, centralized open market, local market, and hybrid), each with different trade-offs in cost, lead time, and compliance burden
- Early planning at trial design stage, rigorous supplier qualification, and robust expiry management consistently determine success
- Working with an FDA-registered distributor like VuRoyal Pharmaceutical — with end-to-end supply chain capabilities and free regulatory guidance — helps sponsors avoid sourcing delays and compliance gaps
Why Comparator Sourcing Is Critical in Global Clinical Trials
The comparator drug must be pharmacologically equivalent to the approved standard of care in each trial region. Mismatches in formulation, strength, or market authorization between regions can render trial results inconclusive or non-transferable to regulatory submissions, invalidating months or years of work and investment.
Patient Safety and Ethics
Sourcing comparators from unaudited or unverified suppliers introduces risk of counterfeit, substandard, or improperly stored product reaching trial participants. The WHO estimates 1 in 10 medicines in low- and middle-income countries fail quality control tests. This failure carries both ethical and liability consequences that can terminate trials and damage sponsor reputations.
Regulatory Compliance Pressure
Global trials must satisfy Good Manufacturing Practice (GMP) and Good Distribution Practice (GDP) standards in every country where the trial operates — not just one. Failure at even one site can trigger authorization delays or complete termination.
Each regulatory market imposes unique documentation and compliance burdens:
- FDA (US): Drug Supply Chain Security Act (DSCSA) traceability requirements
- EMA (EU): Qualified Person certification for each imported batch
- NMPA (China): Pre-approval batch import rules and in-country registration
The Financial Stakes
Clinical trial sponsors spend an average of $50 million per year on clinical supplies, with half of this budget allocated to comparator drugs and co-therapies. According to a 2024 Tufts Center study, a single day of delayed market entry now costs approximately $800,000 in unrealized prescription sales, plus $40,000 in direct daily trial costs.
Supply Chain Reliability
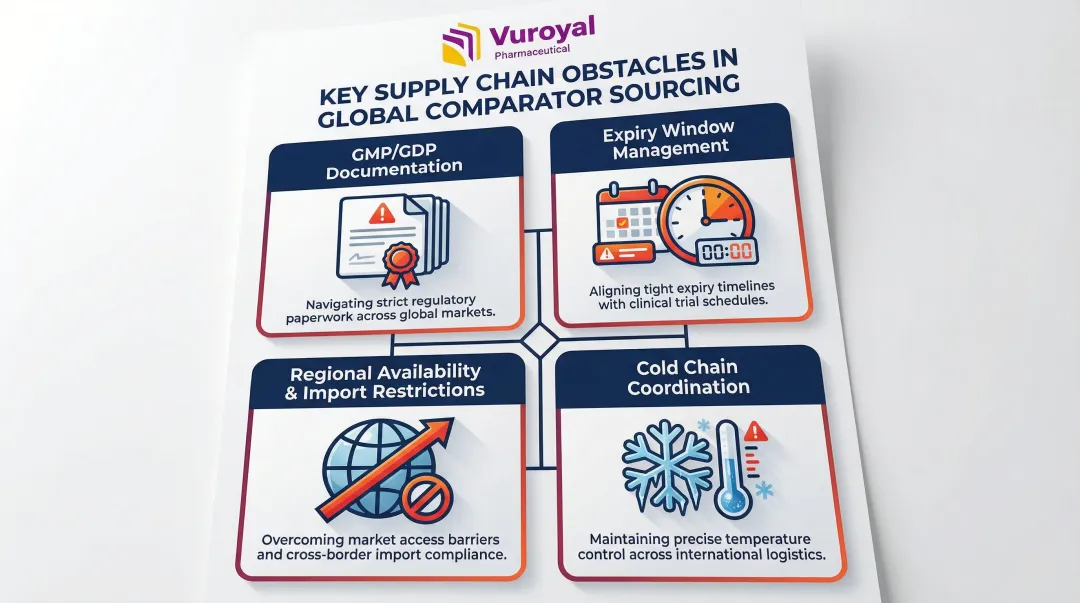
Over 60% of sponsors identify comparator sourcing as a key stumbling block causing time and budget losses. The most persistent obstacles are:
- Obtaining complete GMP/GDP documentation from manufacturers
- Managing short expiry windows against multi-site trial timelines
- Navigating regional availability constraints and import restrictions
- Coordinating cold chain logistics across multiple regulatory jurisdictions
Addressing these challenges requires a sourcing strategy built into trial design — not patched in after supply failures emerge.
How Comparator Sourcing Works: From Planning to Delivery
Comparator sourcing in global trials spans the full lifecycle of a study—from trial design through clinical site delivery, inventory management, and resupply cycles. Each phase builds on the last, and gaps at any stage can delay enrollment or compromise trial integrity.
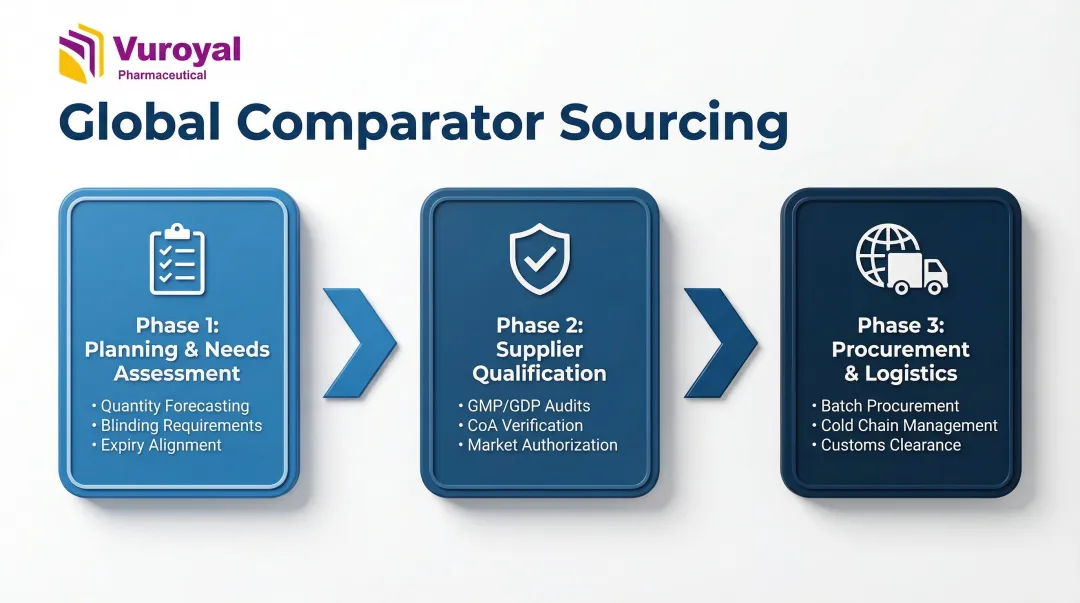
Step 1: Early Planning and Needs Assessment
Sponsors should initiate the comparator sourcing conversation during the trial's protocol design phase—not after site activation. This early planning covers:
- Required quantities per country based on enrollment projections and dropout assumptions
- Blinding and matching requirements such as placebo matching, over-encapsulation, or relabeling needs
- Expiry date requirements aligned to planned packaging campaigns and patient dosing timelines
- Co-medication and supporting supply needs that must be sourced alongside the primary comparator
Early engagement enables better strategy selection, avoids expiry date conflicts, and accounts for lead times that can stretch 6–12 months for manufacturer-direct sourcing or specialty products. Early engagement enables better strategy selection, avoids expiry date conflicts, and accounts for lead times stretching 6–12 months for manufacturer-direct or specialty products. With sourcing timelines confirmed, the next step is qualifying the suppliers who will fulfill them.
Step 2: Supplier Identification, Qualification, and Documentation
The supplier qualification process involves:
- Auditing suppliers for GMP/GDP compliance through on-site inspections or third-party audit reports
- Verifying Certificates of Analysis (CoA) for each batch to confirm product specifications
- Assessing market authorization status in each trial country to ensure regulatory acceptability
- Building documentation such as equivalency statements, pharmaceutical quality comparisons, and regulatory justifications needed when sourcing across different regulatory markets
Once suppliers are qualified, procurement and logistics must be coordinated to move compliant product across borders without delays.
Step 3: Procurement, Packaging, Logistics, and Compliance Clearance
The procurement phase involves:
- Securing product from manufacturer or wholesaler with appropriate batch sizes and expiry windows
- Coordinating relabeling or blinding operations to maintain trial integrity
- Managing cold chain logistics for temperature-sensitive comparators—the pharma industry loses an estimated $35 billion annually due to cold chain failures
- Handling country-specific import/export clearance, customs documentation, and in-country regulatory filings before product reaches clinical sites
Coordinating these activities across multiple countries simultaneously is operationally demanding. Most sponsors rely on either highly resourced internal teams or specialized external partners with established global reach to execute this phase without delays.
Comparator Sourcing Strategies for Global Trials
No single sourcing strategy fits all trials. The right model depends on the trial's geographic footprint, the specific comparator, timeline constraints, blinding requirements, and budget. Many global trials use a combination of approaches at different phases.
Direct from Manufacturer
Pros:
- Fewer supply chain links reduce tampering and counterfeiting risk
- Sponsors can negotiate large single batches with maximum expiry windows
- Certificate of Analysis documentation is readily accessible
- Best batch consistency for blinded trials
Cons:
- Manufacturers typically require trial information before agreeing to supply
- Pricing tends to be higher, aligned to clinical trial pricing rather than wholesale
- Lead times are longer if additional manufacturing campaigns are needed
- Manufacturers may decline to supply competitors' trials
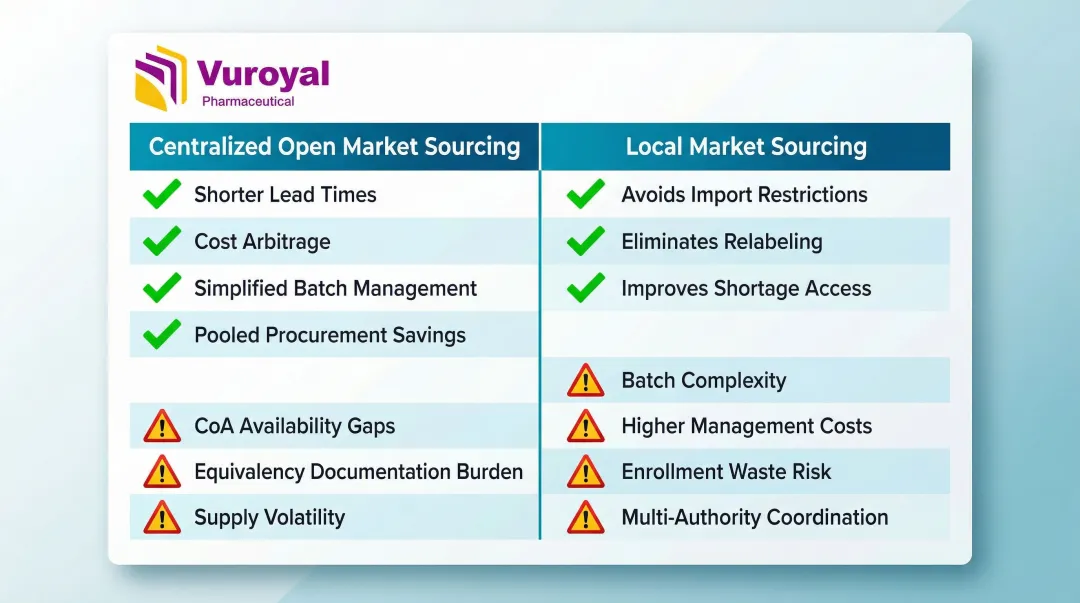
Centralized Open Market Sourcing
Centralized sourcing accounts for approximately 70% of studies requiring comparators, representing the dominant approach for global trials.
Pros:
- Product may already be released and available in wholesaler inventory, enabling shorter lead times
- Sponsors can exploit drug price differentials across countries to reduce costs
- Centralizing procurement in one market simplifies batch management, expiry tracking, and QA/QP release
- Studies show pooled procurement yields price decreases in 79% of cases versus 30% for segmented approaches
Cons:
- Certificates of Analysis may not always be available from wholesalers
- Products sourced from foreign markets for use in different regulatory regions may require advance discussion with local regulators and supporting equivalency documentation
- Supply can be volatile if demand outpaces wholesaler quotas
Local Market Sourcing
Pros:
- Avoids the need for import authorization in countries where importing foreign medicines is restricted
- For open-label trials, local sourcing can eliminate relabeling and patient information leaflet translation requirements
- Sourcing small quantities from multiple markets can improve access during periods of shortage
Cons:
- Managing multiple markets simultaneously increases batch complexity and expiry date management burden
- Higher setup, storage, and management fees compared to centralized models
- If a recruited country underperforms on enrollment, locally sourced product may be wasted
- Requires coordination with multiple local distributors and regulatory authorities
Hybrid Approaches and Partner Selection
Hybrid strategies—such as using open market sourcing to launch a trial quickly while awaiting manufacturer-supplied inventory for later phases—allow sponsors to balance speed, cost, and supply reliability across multi-year global trials.
Executing a hybrid strategy across multiple geographies requires either a highly resourced internal team or a specialized external partner. VuRoyal Pharmaceutical, an FDA-registered wholesaler based in Boston, handles imported drug approval registration, customs clearance, and cold chain logistics for sponsors sourcing FDA Orange/Purple Book reference drugs across international markets — including free regulatory guidance — so teams aren't managing each country's requirements independently.
Key Factors That Affect Global Comparator Sourcing
Regulatory Divergence
Each country has its own requirements for drug registration, import licensing, labeling language, and GMP equivalency recognition. What satisfies one regulator may require additional documentation in another. For example, Japan requires a "Yakkan" certificate for importing unapproved drugs, while the EU requires physical importation and Qualified Person certification before batch release.
Drug Pricing Variation
The same comparator can vary significantly in list price across national markets. Lower-cost markets offer savings, but they often carry limited stock and higher supply risk. Backup sourcing plans and ongoing market intelligence are essential to managing this trade-off.
Expiry Date Management
Open-market stock frequently carries shorter expiry windows than manufacturer-direct supply. Sponsors must align sourcing timelines to packaging campaigns and patient enrollment projections to avoid waste or emergency resupply. Mid-study expiry events can halt enrollment and trigger costly protocol amendments.
Cold Chain and Special Handling Requirements
Biologics comprise 46% of total medicine spending in the US, and biosimilar comparators are increasingly common in clinical trials. These products require temperature-controlled logistics throughout the supply chain. The WHO notes that up to 50% of vaccines are wasted globally each year due to inadequate temperature control. A single cold chain failure can invalidate product and shut down a trial entirely.
Counterfeit and Supply Integrity Risk
Multi-tier wholesaler networks and lower-regulation markets introduce real traceability risk. Between August 2017 and December 2021, the WHO recorded 877 incidents of substandard and falsified medical products — an average annual increase of 36.3%. Sponsors sourcing comparators globally should require:
- Regular supplier audits with documented qualification records
- Serialization tracking across all distribution tiers
- Independent testing programs for incoming product batches

Common Challenges and Mistakes to Avoid
Common Challenges and Mistakes to Avoid
Starting Too Late
Treating comparator sourcing as a late-stage procurement task—rather than integrating it into trial design and budget planning—consistently leads to supply shortages, protocol deviations, and avoidable cost escalation. Mid-study sourcing attempts limit options and inflate costs because suppliers have less inventory flexibility and sponsors have fewer alternatives.
Assuming Market Availability Without Intelligence
Sponsors often assume that a commercially approved comparator will be easily available in every trial country. In practice, export bans, manufacturer quotas, drug shortages, and local regulatory restrictions can make supply unexpectedly difficult. A proper market assessment before trial start prevents this and identifies backup suppliers early.
Underestimating Documentation and Equivalency Requirements
Cross-market sourcing — such as EU-sourced product for a US trial or US-sourced product for China — requires more than just finding available supply. Sponsors must prepare equivalency documentation and engage regulators before procurement begins.
Health Canada's guidance, for example, requires proof that foreign-sourced reference products are identical in color, shape, size, weight, and coating, along with Certificates of Analysis and proof of purchase. Minimum requirements like these vary by jurisdiction. Missing them can trigger batch rejection, IND submission delays, or a full trial hold.
Regulatory documentation for comparators is rarely just a formality. Build it into your sourcing timeline from the start.
Conclusion
Comparator sourcing in global clinical trials is a strategic supply chain discipline that spans regulatory compliance, supplier qualification, logistics management, and cost optimization—not a simple purchasing task. It directly determines whether a trial runs on time and produces credible results that regulators will accept.
The right strategy—whether centralized, local, hybrid, or direct—depends on the specific trial's requirements, geographic footprint, and timeline constraints. Sponsors who engage sourcing partners with deep regulatory knowledge, established supplier networks, and end-to-end logistics capabilities are best positioned to avoid the delays and cost overruns that derail comparative studies.
VuRoyal Pharmaceutical is an FDA-registered, Massachusetts-licensed pharmaceutical wholesaler built specifically for clinical trial sponsors navigating international procurement. Core capabilities include:
- Reference Listed Drug (RLD) sourcing for generic development and comparative studies
- Cold chain logistics with end-to-end temperature-controlled transportation
- Customs clearance and import facilitation for cross-border shipments
- Free regulatory guidance on imported drug approval, registration, and filing requirements
For sponsors managing complex multi-country trials, VuRoyal offers a single point of contact—from initial procurement through final site delivery. Contact their team at +1 617-870-2690 to discuss sourcing requirements.
Frequently Asked Questions
What is comparator sourcing in clinical trials?
Comparator sourcing is the process of identifying, qualifying, and procuring approved reference drugs used in comparative clinical trials. It requires careful planning across regulatory, supply chain, and documentation dimensions to ensure products meet GMP/GDP standards and regulatory equivalency requirements in every trial country.
What is a comparator in a clinical trial?
A comparator (also called a reference drug) is a commercially approved medication used alongside or in place of a placebo to measure whether an investigational drug performs better than the current standard of care. Comparators establish the efficacy and safety baseline against which new treatments are evaluated.
What is the difference between central and local comparator sourcing?
Centralized sourcing involves procuring, packaging, and labeling from a single country for global distribution, offering cost efficiency and simplified batch management. Local sourcing procures from within each trial country, avoiding import restrictions but increasing inventory complexity and setup costs across multiple markets.
How early should comparator sourcing planning begin?
Sourcing planning should begin at the trial protocol design stage, not after site activation. Early engagement avoids expiry date conflicts, accounts for 6–12 month lead times on specialty products, and keeps co-medication and ancillary supply timelines aligned.
What are the main risks in global comparator sourcing?
Primary risks include supply shortages, counterfeit product infiltration, regulatory non-compliance across jurisdictions, cold chain failures for biologics (causing up to $35 billion in annual pharma losses), and expiry date mismanagement.
How do GMP and GDP requirements apply to comparator sourcing?
Comparator drugs must be sourced from GMP-compliant manufacturers and distributed through GDP-compliant channels in every trial region. Certificates of Analysis, batch records, and audit reports must be maintained throughout the supply chain to satisfy regulators and confirm product integrity at every handoff.


