
Introduction
In healthcare, a delayed shipment isn't just an inconvenience—it can halt clinical trials, compromise drug efficacy, or trigger a regulatory crisis. When a temperature-sensitive vaccine adjuvant arrives outside specification, or when a critical reference drug is detained at customs, the consequences ripple through research timelines, patient access, and compliance records.
These aren't hypothetical scenarios. They're daily realities for pharmaceutical distributors, hospitals, research organizations, and supply chain teams operating under the most tightly regulated logistics requirements of any industry.
Healthcare logistics management operates under stricter requirements than any other supply chain sector. A single temperature excursion can render a $50,000 shipment unusable. Missing documentation can trigger FDA import refusals. With the biopharma industry losing an estimated $35 billion annually to temperature-controlled logistics failures, the pressure to get every link in the chain right is relentless.
This guide covers what healthcare logistics management involves, the unique challenges that make it complex, and the best practices that help supply chain teams optimize operations from procurement to point of care.
TLDR
- Pharmaceutical logistics operates under strict regulatory frameworks (FDA, GDP, GMP) — compliance failures can halt distribution and jeopardize patient care
- Cold chain products now represent 32% ($384 billion) of global pharmaceutical value, growing 13% annually
- U.S. hospitals spent 20 million hours managing drug shortages in 2023, costing $900 million in labor alone
- 80% of health systems lack real-time supply chain visibility, leaving them unable to prevent disruptions before they occur
- Demand forecasting, supplier diversification, and IoT-enabled cold chain monitoring are the highest-impact levers for supply chain resilience
What Is Healthcare Logistics Management?
Healthcare logistics management is the coordinated planning, execution, and control of the movement, storage, and delivery of pharmaceuticals, medical devices, biological specimens, and healthcare supplies from manufacturers to healthcare providers and patients. The core requirement is non-negotiable: the right product must arrive in the right condition, at the right place, at the right time.
Unlike general logistics, healthcare logistics operates under strict regulatory frameworks across multiple jurisdictions. Products are often temperature-sensitive and expiry-sensitive, with life-critical delivery timelines. Many require specialized handling for hazardous or controlled substances.
Regulatory oversight spans several major bodies: the FDA's 21 CFR Part 211 in the United States, the European Medicines Agency's Good Distribution Practice guidelines, and the WHO's Model Guidance for time- and temperature-sensitive pharmaceutical products.
Four functional pillars form the backbone of healthcare logistics:
- Procurement and sourcing: Qualify suppliers, manage regulatory documentation, and secure hard-to-source specialty products
- Transportation: Manage cold chain integrity, customs clearance, and in-transit monitoring for pharmaceutical shipments
- Warehousing and storage: Operate temperature-controlled facilities with inventory rotation protocols and compliance documentation
- Distribution: Execute last-mile delivery to hospitals, clinics, research sites, and pharmacies
Each pillar carries its own compliance burden — and failure in any one area can disrupt the entire supply chain. That operational complexity helps explain the scale of the market: global pharmaceutical logistics reached $99.33 billion in 2024 and is projected to grow to $208.26 billion by 2033, driven largely by the expansion of biologics and specialty pharmaceuticals requiring advanced cold chain infrastructure.

Core Challenges in Healthcare Logistics Operations
Regulatory Complexity Across Jurisdictions
Healthcare logistics must comply with overlapping regulatory frameworks that vary by product type, destination country, and transport mode. Two of the most demanding examples:
- FDA 21 CFR 211.142 requires written procedures for warehousing, quarantine protocols, and storage under appropriate temperature and humidity conditions
- EU GDP guidelines mandate temperature mapping of storage areas, calibrated monitoring equipment, and alarm systems for temperature excursions
Non-compliance carries severe consequences. The FDA actively penalizes manufacturers and distributors through Warning Letters that can result in shipment seizures, import rejections, product recalls, and patient harm. In February 2025, the FDA issued a Warning Letter to ABR Laboratory LLC after investigators found a 2–8°C refrigerator reached 17.4°C for over 24 hours—a deviation that was neither identified nor investigated, compromising reference microorganisms. Similarly, Atlantic Management Resources received a Warning Letter in January 2023 for failing to monitor temperature and humidity for bulk drugs sensitive to heat and moisture, resulting in a production halt and nationwide recall.
For organizations importing specialty drugs or pharmaceutical excipients—particularly complex biologics like vaccine adjuvants—manual documentation management is error-prone and resource-intensive. Required records include certificates of analysis, import licenses, customs declarations, and chain-of-custody files. Cross-border operations carry significantly higher compliance risk without dedicated regulatory support.
Temperature Sensitivity and Cold Chain Failures
Cold chain medicines now account for 32% ($384 billion) of global pharmaceutical value, growing at 13% annually—more than four times the 3% growth rate of ambient temperature medicines. This shift reflects the expansion of biologics, vaccines, and specialty pharmaceuticals that require strict temperature control throughout storage and transit.
Yet temperature excursions remain a persistent problem. A 2023 study published in the Journal of the American Pharmacists Association found that simulated mail-transit packages spent an average of 68.3% of transit time outside the USP-recommended range of 68°F to 77°F. Even brief excursions can render products unsafe or ineffective, with consequences ranging from wasted inventory to patient harm.
The biopharma industry loses approximately $35 billion annually due to failures in temperature-controlled logistics, according to the IQVIA Institute for Human Data Science. That figure encompasses lost product, clinical trial replacement costs, wasted logistics expenses, and the labor required for root-cause analysis.
Drug Shortages and Unpredictable Demand
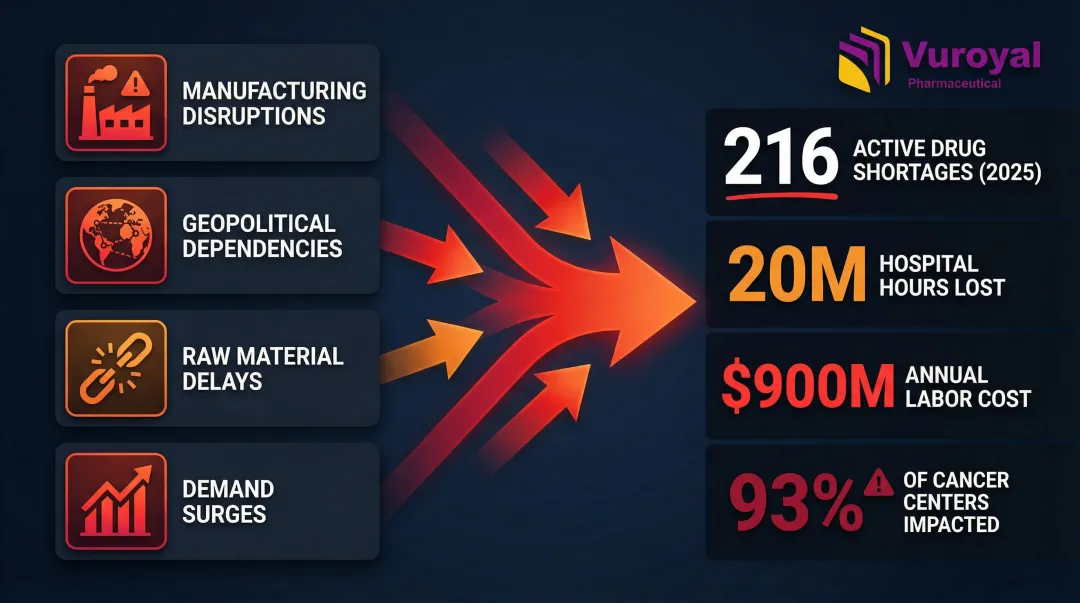
The U.S. healthcare system is navigating a persistent drug shortage crisis. According to the American Society of Health-System Pharmacists (ASHP), there were 216 active drug shortages in early 2025, down slightly from 271 at the end of 2024. But the numbers don't capture the operational burden: U.S. hospitals spent roughly 20 million hours managing drug shortages in 2023, translating to nearly $900 million annually in labor costs—more than double the $360 million reported in 2019.
The root causes are diverse:
- Manufacturing disruptions — Quality failures at single-source facilities
- Geopolitical supply dependencies — India's 2020 export restrictions on critical APIs led to a 37% increase in new U.S. shortage listings within six weeks
- Raw material delays — Geographic concentration of API production
- Demand surges — Disease outbreaks or new treatment introductions
The downstream impact forces organizations to seek expensive alternatives, reschedule procedures, or maintain costly backup inventory. The 2023 cisplatin and carboplatin shortages illustrate the stakes: a single manufacturer's quality failure impacted 93% of U.S. cancer centers and delayed treatments for 16% of patients.
Fragmented Systems and Limited Supply Chain Visibility
Supply chain visibility remains a critical blind spot. A January 2026 national survey by Tecsys revealed that 80% of health systems operate without real-time visibility, relying instead on delayed, partial, or manual tracking. Only 20% of healthcare leaders reported having full, real-time visibility across their inventory and care settings.
Fragmented data systems prevent logistics teams from tracking shipments proactively, identifying bottlenecks, responding to delays, or establishing meaningful performance benchmarks. Without real-time visibility, organizations cannot intercept temperature excursions in transit—they discover failures after delivery, forcing them to scrap degraded products rather than intervene proactively.
McKinsey research indicates that once companies experience a supply chain disruption, it takes an average of two weeks to plan and execute a response. In pharmaceutical logistics, where products may have hours-long stability windows outside specification, this delay is unacceptable.
Best Practices for Optimizing Healthcare Logistics Management
Centralize Data and Build End-to-End Supply Chain Visibility
The foundation of effective healthcare logistics is a unified, real-time view of inventory levels, shipment status, and supplier performance. Standardizing product data across systems eliminates silos that prevent logistics teams from seeing the complete picture.
Key steps include:
- Implementing integrated dashboards that consolidate data from procurement, warehouse management, transportation, and distribution systems
- Establishing automated alerts for inventory thresholds, shipment delays, and temperature excursions
- Creating supplier scorecards that track on-time delivery, quality metrics, and compliance performance
- Deploying track-and-trace systems that provide location and condition data throughout transit
Centralized visibility lets logistics teams anticipate disruptions before they escalate, cutting costly delays, preventing stockouts, and strengthening compliance documentation.
Implement Demand Forecasting and Inventory Controls
Accurate, data-driven demand forecasting allows healthcare organizations to right-size inventory, reducing both stockouts and waste. Using historical usage data, seasonal patterns, and machine learning models, logistics teams can predict demand with greater precision.
For expiry-sensitive pharmaceutical products, strict inventory rotation protocols are essential:
- FEFO (First Expired, First Out) — The WHO explicitly endorses this method for time- and temperature-sensitive pharmaceutical products
- FIFO (First In, First Out) — Used when expiration dates are uniform or products are not expiry-sensitive
Poor inventory management results in significant financial losses. A recent case study found that expired drugs made up approximately 8% of a hospital's total inventory value before intervention. Structured oversight brought that figure down to 5.2%, moving closer to the industry benchmark of 3%.
Globally, the WHO and UNEP estimate that up to 50% of vaccines are wasted annually, largely due to insufficient logistics and temperature control.
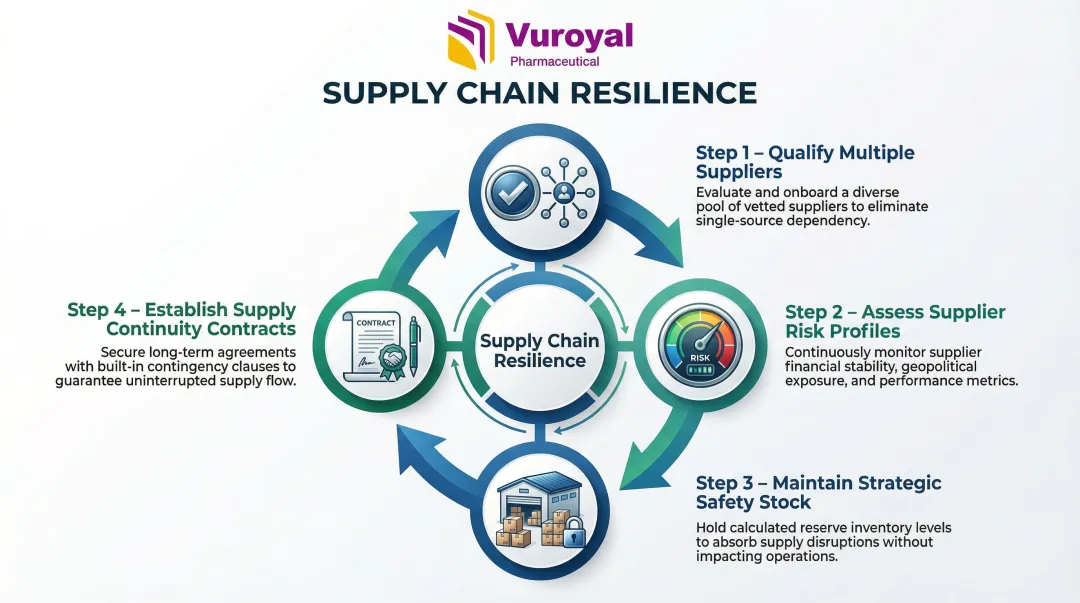
Diversify Supplier Relationships and Reduce Single-Source Dependency
Relying on a single supplier—especially for specialty drugs, imported biologics, or critical vaccine components—creates fragile supply chains. The 2023 oncology shortage demonstrated this risk: a single manufacturer's facility closure caused severe cisplatin shortages, impacting 93% of U.S. cancer centers.
Supplier diversification strategies include:
- Qualifying multiple suppliers for high-criticality items
- Assessing each supplier's risk profile (geographic concentration, production capacity, financial stability)
- Maintaining strategic safety stock for products with long lead times or single-source vulnerabilities
- Establishing contractual agreements that include supply continuity clauses
For organizations sourcing specialty pharmaceuticals or imported products, partnering with an FDA-registered pharmaceutical distributor with international procurement experience—such as VuRoyal Pharmaceutical—can provide access to diversified supply networks and regulatory expertise that would be difficult to build in-house.
Standardize Processes and Implement SOPs Across the Supply Chain
Standard operating procedures for receiving, storage, dispensing, returns, and disposal reduce human error across facilities and staff rotations. SOPs deliver value only when paired with consistent cross-functional training, clear accountability, and regular process audits.
Critical SOP areas include:
- Temperature monitoring and documentation protocols
- Quarantine and release procedures for incoming shipments
- Stock rotation and expiry management
- Deviation investigation and corrective action processes
- Equipment calibration and maintenance schedules
Organizations operating under GDP guidelines must anchor their SOPs to applicable regulatory frameworks, including:
- USP guidelines
- Title 21 CFR
- Prescription Drug Marketing Act (PDMA)
- Drug Supply Chain Security Act (DSCSA)
Third-party compliance auditors provide independent verification that SOPs are followed consistently across sites and staff rotations.
Build Supply Chain Resilience Through Scenario Planning and Risk Management
Building resilience starts with mapping supply chain vulnerabilities—identifying which products, routes, and suppliers carry the highest risk—and documenting contingency plans before disruptions occur.
Scenario planning exercises should address:
- Supplier facility shutdowns or quality failures
- Transportation disruptions (weather events, geopolitical conflicts)
- Regulatory changes or import restrictions
- Demand surges from disease outbreaks or new treatment launches
Regular stress-testing reveals gaps before they become crises. Contingency plans should specify pre-qualified alternate suppliers, backup transportation routes, and safety stock calculations tied to lead time variability.
Optimize Network Design and Distribution Routes
Geographic placement of suppliers, distribution centers, and healthcare facilities directly affects delivery speed and cost. Evaluating transportation modes, carrier performance, and last-mile delivery options together produces more reliable outcomes than optimizing any one factor in isolation.
For specialty or temperature-sensitive shipments, route optimization is not just a cost exercise but a product integrity requirement. Factors to consider include:
- Transit time and temperature stability windows
- Availability of refrigerated transportation (reefers)
- Customs clearance efficiency at border crossings
- Last-mile delivery capabilities to remote or rural facilities
Advanced logistics modeling can identify optimal distribution center locations, consolidate shipments to reduce handling, and guide carrier selection based on performance data alongside cost.
Cold Chain and Specialty Pharmaceutical Logistics
The Temperature Spectrum for Pharmaceutical Cold Chains
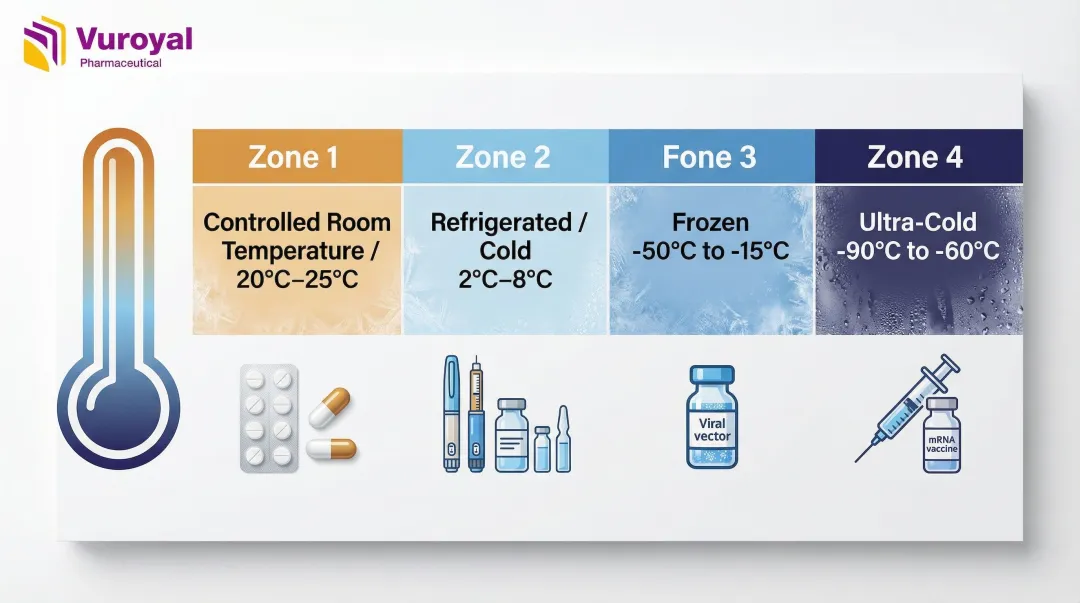
Pharmaceutical cold chains must manage a precise temperature spectrum, with different products requiring different conditions:
| Classification | Temperature Range | Regulatory Definition |
|---|---|---|
| Controlled Room Temperature (CRT) | 20°C to 25°C | USP <1079.2> permits transient excursions between 15°C and 30°C, provided Mean Kinetic Temperature is managed |
| Refrigerated (Cold) | 2°C to 8°C | Standard for most biologics, insulins, and traditional vaccines |
| Frozen | -50°C to -15°C | Required for specific viral vectors and vaccines |
| Ultra-Cold | -90°C to -60°C | Required for mRNA vaccines and advanced cell therapies |
Many specialty biologics—including vaccines and their adjuvants—require precise maintenance within these narrow ranges. Any excursion, even briefly, can compromise product safety, efficacy, and regulatory status. Downstream handlers typically lack the stability data required to justify deviations, making excursion prevention critical.
Storage and Packaging Infrastructure Requirements
Certified cold chain logistics requires appropriate storage facilities (walk-in cold rooms, ultra-low temperature freezers) and specialized packaging solutions.
Passive containers use insulated materials with coolants like dry ice or gel packs. The International Safe Transit Association (ISTA) provides Standard 20 and 7E profiles for qualifying passive Insulated Shipping Containers (ISCs). Both profiles are suited to shorter transit durations and products with wider temperature tolerances.
Active containers use electrically controlled cooling with integrated temperature sensors. The Parenteral Drug Association (PDA) provides Technical Report 64 for qualifying active temperature-controlled systems. Active systems are required for longer transit durations, products with narrow temperature windows, or high-value shipments where any risk is unacceptable.
Choosing between passive and active systems depends on product sensitivity, transit duration, transport mode, and cost. For ultra-cold products like certain mRNA vaccines, active containers with backup power systems are often the only viable option.
Transport Requirements and In-Transit Condition Monitoring
Cold chain integrity during transport demands refrigerated vehicles (reefers), validated packaging for specific route durations, and real-time IoT-enabled monitoring that sends automated alerts when temperature deviations occur.
WHO Annex 9 mandates that temperature records for time- and temperature-sensitive pharmaceutical products be maintained with a minimum recording frequency of six times per hour for each monitoring sensor position. EU GDP guidelines require that monitoring equipment be maintained and calibrated at regular intervals.
Data logging throughout transit is not only operationally necessary — it is a regulatory requirement. Every shipment must carry documented evidence of temperature conditions at each stage, demonstrating unbroken cold chain integrity from manufacturer to end user.
For organizations sourcing specialty drugs or imported pharmaceutical products—including complex biologics like vaccine adjuvants—this documentation burden is substantial. VuRoyal Pharmaceutical, an FDA-registered wholesaler, provides dedicated cold chain transportation and certified end-to-end supply chain capabilities, handling temperature-sensitive logistics, regulatory compliance, and customs clearance while delivering full chain-of-custody documentation.
Technology and Compliance in Modern Healthcare Logistics
IoT Sensors and Real-Time Condition Monitoring
The industry is rapidly moving away from passive data loggers toward Internet of Things (IoT) telemetry. Analysts predict that 75% of all pharmaceutical shipments will use IoT-based tracking by 2030.
IoT infrastructure with Bluetooth or cellular sensors attached to storage units and shipping containers enables continuous monitoring of temperature, humidity, and location. Automated deviation alerts allow logistics teams to intercept shipments, reroute to alternate carriers, or notify recipients to quarantine products before failures reach the point of delivery.
IoT platforms also generate the immutable audit trails and secure record retention required by FDA 21 CFR Part 11. EU GDP guidelines separately mandate alarm systems for temperature excursions — IoT infrastructure automates that alert process while simultaneously producing compliance documentation.

Inventory Management and Quality Management Software
Digital inventory tools handle the operational complexity of pharmaceutical stock management. Core capabilities include:
- Automating FEFO/FIFO workflows to enforce proper stock rotation
- Tracking lot numbers, expiration dates, and storage locations in real time
- Generating reorder alerts before stockouts occur
- Preventing dispensing errors tied to expired or misidentified products
Quality Management Systems (QMS) extend this further by managing supplier qualifications, documenting standard operating procedures, tracking regulatory changes, and maintaining audit-ready records.
Advanced systems integrate with procurement, warehouse management, and transportation platforms, providing end-to-end visibility and automated compliance documentation across the entire supply chain.
Cross-Border Pharmaceutical Logistics and Regulatory Compliance Support
Cross-border pharmaceutical imports carry layered compliance requirements that compound quickly. A single shipment of specialty drugs or novel excipients may require:
- FDA export documentation and customs declaration filings
- Import registration processes in the destination country
- Destination-country regulatory approvals specific to the product class
The U.S. Drug Supply Chain Security Act (DSCSA) adds another layer by mandating an interoperable electronic system to identify and trace prescription drugs at the package level. Manufacturers, distributors, and dispensers must capture and maintain full transaction histories — a serialization requirement that significantly increases data complexity for both domestic and cross-border logistics.
The FDA's Import Refusal Report (IRR) tracks shipments denied entry due to compliance failures, including improper storage or adulteration. Organizations without specialized expertise in pharmaceutical imports face significant risk of border detentions, shipment rejections, and regulatory penalties.
Working with a certified importer that specializes in international pharmaceutical procurement reduces this exposure considerably. VuRoyal Pharmaceutical, for example, manages the complete import lifecycle — from approval registration through customs clearance and final delivery — with full regulatory compliance and documented chain of custody built into every shipment.
Frequently Asked Questions
What does healthcare logistics do?
Healthcare logistics manages the movement, storage, and delivery of medical supplies, pharmaceuticals, and equipment from manufacturers to healthcare facilities and patients. It ensures the right products arrive in the right condition, at the right time, to support patient care and clinical research.
What are the 4 types of logistics management?
The four core types are procurement (sourcing and supplier management), transportation (shipment and delivery), warehousing and inventory management (storage and stock control), and last-mile distribution (final delivery to end users). In healthcare, each requires specialized expertise to meet regulatory requirements and protect product quality.
What are the 7 C's of logistics?
The 7 C's are the right Customer, right Commodity, right Quantity, right Condition, right Place, right Time, and right Cost. In healthcare logistics, each carries especially high stakes because failures can directly impact patient safety and treatment outcomes.
What is cold chain management in healthcare logistics?
Cold chain management is the process of maintaining required temperature conditions for temperature-sensitive pharmaceutical products, including vaccines, biologics, and specialty drugs, throughout storage, packaging, and transportation. It requires validated equipment, real-time monitoring, and documented protocols to keep products within specification.
How does regulatory compliance affect healthcare logistics operations?
Regulatory frameworks like FDA guidelines, GDP, and GMP impose strict requirements on how pharmaceutical products are stored, transported, and documented. Compliance demands specialized facilities, validated processes, and continuous monitoring — all of which add operational complexity but ultimately protect product quality and patient safety.
What should healthcare organizations look for in a pharmaceutical distribution partner?
Look for FDA registration, cold chain transportation capabilities, a strong compliance track record, and international procurement experience. The right partner goes beyond shipping — providing chain-of-custody documentation, customs clearance support, regulatory guidance, and end-to-end supply chain management.


