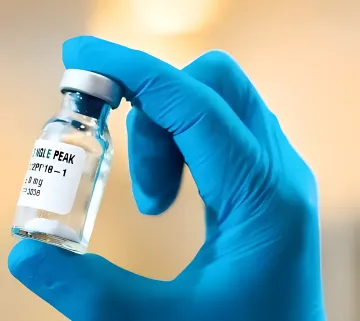
QS-21 Human Vaccine Adjuvant
- Used in 5 globally approved vaccines
- 200–400 million human doses annually
- EXCiPACT & IPEC GMP certified quality




GMP Certified
IPEC GMP Certified
FDA-Registered Distributor
This category covers vaccine adjuvant manufacturers and companies worldwide, featuring GMP-certified, clinically validated adjuvants such as QS-21 derived from Quillaja saponaria. Products serve human vaccine development, veterinary applications, and clinical research, enhancing both antibody production and cellular immunity across approved vaccines for shingles, malaria, RSV, and COVID-19.
Talk to our experts for custom solutions and tailored guidance.
Apply Filters





Several adjuvants are currently used in approved vaccines. QS-21, a saponin derived from Quillaja saponaria, is used in Shingrix (shingles), Arexvy (RSV), Mosquirix and R21/Matrix-M (malaria), and COVID-19 vaccines. Other common adjuvants include aluminum salts (alum), MF59 (squalene-based), AS04 (alum + MPL), and Matrix-M. QS-21 is often incorporated into composite adjuvant systems such as AS01, AS02, and ALFQ.
Talk to our experts for custom solutions and tailored guidance.

QS-21 adjuvant powers approved vaccines for shingles, RSV, malaria, and COVID-19, and supports 20+ clinical trials.

Supports clinical trial sponsors with QS-21 supply, reference listed drugs, and end-to-end regulatory compliance services.

Saponin-based adjuvants used in poultry, swine, aquaculture, and ruminant vaccination programs to boost immunity.

FDA-registered distribution of adjuvants and reference drugs to hospitals, clinics, and research institutions worldwide.
Talk to our experts for custom solutions and tailored guidance.
As Desert King International's exclusive China partner since 2023, VuRoyal provides direct access to QS-21 used in 5 globally approved vaccines.
From imported drug approval registration to cold chain logistics and customs clearance, VuRoyal manages the full supply chain with free regulatory guidance.
Founded by pharmaceutical R&D and clinical research professionals, VuRoyal ensures safe, genuine, GMP-certified adjuvants and reference drugs for your research.
Submit your inquiry and our specialists will respond with pricing, regulatory guidance, and supply chain options tailored to your research needs.
For immediate assistance, feel free to give us a direct call at +1 617-870-2690 You can also send us a quick email at
For immediate assistance, feel free to give us a direct call at +1 617-870-2690 You can also send us a quick email at