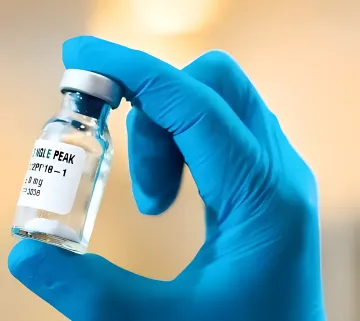
QS-21 Human Vaccine Adjuvant
- Used in 5 globally approved vaccines
- 20,000–40,000 human doses per gram
- EXCiPACT & GMP certified supply chain


GMP Certified
EXCiPACT Certified
FDA-Registered Distributor
Hepatitis B vaccines with adjuvants rely on potent immunological agents like QS-21, a GMP-certified saponin derived from Quillaja saponaria Molina, to enhance both antibody production and cellular immunity. Used in clinical trials and approved vaccine formulations globally, these adjuvants support the development of next-generation hepatitis B and other infectious disease vaccines.



Hepatitis B vaccine candidates in clinical research have utilized QS-21, a highly purified saponin derived from Quillaja saponaria Molina bark. QS-21 enhances both antibody production and cellular immunity, making it suitable for hepatitis B and other infectious disease vaccine formulations. It is also a key component in composite adjuvant systems such as AS01 and AS02 used across multiple approved vaccines.
Talk to our experts for custom solutions and tailored guidance.

QS-21 adjuvants support hepatitis B, HIV, cancer, and infectious disease vaccine R&D programs.

Supplies reference-grade QS-21 adjuvant for Phase I–III clinical trials across multiple vaccine candidates.

GMP-certified QS-21 integrated into composite adjuvant systems like AS01 and Matrix-M for commercial vaccine production.

Adjuvant supply supporting vaccines targeting shingles, malaria, RSV, COVID-19, and hepatitis B globally.
Talk to our experts for custom solutions and tailored guidance.
As Desert King International's sole China partner since 2023, VuRoyal provides guaranteed access to the world's leading GMP-certified vaccine adjuvant.
FDA-registered and Massachusetts-approved with free regulatory guidance, import filing, customs clearance, and cold chain logistics for specialty pharmaceutical substances.
Founded by pharmaceutical R&D and clinical research veterans, VuRoyal delivers technical consultation and supply chain support tailored to vaccine innovators worldwide.
Submit your inquiry and our team will respond with pricing, supply details, and free regulatory guidance tailored to your vaccine development program.
For immediate assistance, feel free to give us a direct call at +1 617-870-2690 You can also send us a quick email at
For immediate assistance, feel free to give us a direct call at +1 617-870-2690 You can also send us a quick email at