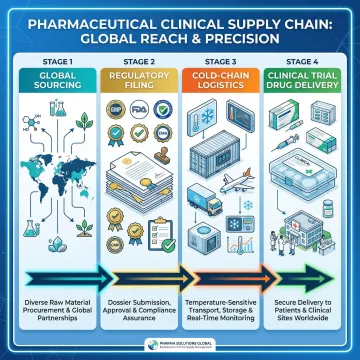
"VuRoyal's Reference Listed Drug service was instrumental for our Phase III trial. Their regulatory guidance ensured seamless FDA compliance, and their cold chain logistics delivered our comparators perfectly preserved. Exceptional reliability."
"We needed QS-21 adjuvants for our vaccine development program urgently. VuRoyal's exclusive partnership with Desert King International gave us access to GMP-certified inventory within weeks. Their expertise in complex regulatory requirements saved us months."
"Custom clinical trial sourcing is notoriously complicated, but VuRoyal's end-to-end supply chain capabilities made our multi-country trial setup painless. Their one-stop procurement and customs clearance solutions were game-changing for our timeline."
"Our hospital needed emergency distribution of specialized pharmaceuticals for our oncology ward. VuRoyal responded within 24 hours with perfect inventory availability and their advanced logistics ensured on-time delivery every single time."
"As a generic drug developer, we appreciate VuRoyal's cost-optimized custom clinical trial sourcing solutions. Their free regulatory guidance on Reference Listed Drugs reduced our approval timeline significantly, and their pricing is competitive."
"We've worked with VuRoyal for three years on multiple clinical trials. Their team's deep pharmaceutical R&D background means they understand our challenges. Their consistency and reliability make them our preferred partner for complex sourcing."
"VuRoyal's specialization in Human Vaccine Adjuvants, particularly their QS-21 GMP inventory, provided exactly what our HIV vaccine research needed. Their technical expertise and access to Desert King's certified facilities was invaluable."
"In the competitive pharmaceutical distribution space, VuRoyal stands apart with FDA registration, Massachusetts licensing, and customs expertise. Their market positioning as an exclusive Desert King partner gives us confidence in supply security and compliance."
"VuRoyal's Reference Listed Drug service was instrumental for our Phase III trial. Their regulatory guidance ensured seamless FDA compliance, and their cold chain logistics delivered our comparators perfectly preserved. Exceptional reliability."
"We needed QS-21 adjuvants for our vaccine development program urgently. VuRoyal's exclusive partnership with Desert King International gave us access to GMP-certified inventory within weeks. Their expertise in complex regulatory requirements saved us months."
"Custom clinical trial sourcing is notoriously complicated, but VuRoyal's end-to-end supply chain capabilities made our multi-country trial setup painless. Their one-stop procurement and customs clearance solutions were game-changing for our timeline."
"Our hospital needed emergency distribution of specialized pharmaceuticals for our oncology ward. VuRoyal responded within 24 hours with perfect inventory availability and their advanced logistics ensured on-time delivery every single time."
"As a generic drug developer, we appreciate VuRoyal's cost-optimized custom clinical trial sourcing solutions. Their free regulatory guidance on Reference Listed Drugs reduced our approval timeline significantly, and their pricing is competitive."
"We've worked with VuRoyal for three years on multiple clinical trials. Their team's deep pharmaceutical R&D background means they understand our challenges. Their consistency and reliability make them our preferred partner for complex sourcing."
"VuRoyal's specialization in Human Vaccine Adjuvants, particularly their QS-21 GMP inventory, provided exactly what our HIV vaccine research needed. Their technical expertise and access to Desert King's certified facilities was invaluable."
"In the competitive pharmaceutical distribution space, VuRoyal stands apart with FDA registration, Massachusetts licensing, and customs expertise. Their market positioning as an exclusive Desert King partner gives us confidence in supply security and compliance."
"VuRoyal's Reference Listed Drug service was instrumental for our Phase III trial. Their regulatory guidance ensured seamless FDA compliance, and their cold chain logistics delivered our comparators perfectly preserved. Exceptional reliability."
"We needed QS-21 adjuvants for our vaccine development program urgently. VuRoyal's exclusive partnership with Desert King International gave us access to GMP-certified inventory within weeks. Their expertise in complex regulatory requirements saved us months."
"Custom clinical trial sourcing is notoriously complicated, but VuRoyal's end-to-end supply chain capabilities made our multi-country trial setup painless. Their one-stop procurement and customs clearance solutions were game-changing for our timeline."
"Our hospital needed emergency distribution of specialized pharmaceuticals for our oncology ward. VuRoyal responded within 24 hours with perfect inventory availability and their advanced logistics ensured on-time delivery every single time."
"As a generic drug developer, we appreciate VuRoyal's cost-optimized custom clinical trial sourcing solutions. Their free regulatory guidance on Reference Listed Drugs reduced our approval timeline significantly, and their pricing is competitive."
"We've worked with VuRoyal for three years on multiple clinical trials. Their team's deep pharmaceutical R&D background means they understand our challenges. Their consistency and reliability make them our preferred partner for complex sourcing."
"VuRoyal's specialization in Human Vaccine Adjuvants, particularly their QS-21 GMP inventory, provided exactly what our HIV vaccine research needed. Their technical expertise and access to Desert King's certified facilities was invaluable."
"In the competitive pharmaceutical distribution space, VuRoyal stands apart with FDA registration, Massachusetts licensing, and customs expertise. Their market positioning as an exclusive Desert King partner gives us confidence in supply security and compliance."